
Research Article
Int J Nutr Sci. 2023; 8(1): 1072.
Phytochemicals Treatment Decreases Drug-Resistant Breast Cancer Stem Cell Viability and Invasiveness by Blocking the HPI/AMF Surviving Pathway
Juan C Gallardo- Pérez1; Jorge L Vargas Navarro2; Diana X Robledo- Cadena1; Silvia C Pacheco-Velázquez1; Joaquín A Padilla Flores2; Rafael Moreno-Sánchez2; Sara Rodríguez- Enríquez3*
¹Departamento de Bioquímica, Instituto Nacional de Cardiología Ignacio Chávez, Ciudad de México, México
²Laboratorio de Control Metabólico, Carrera de Biología, Facultad de Estudios Superiores Iztacala, Universidad Nacional Autónoma de México, Tlalnepantla de Baz, Estado de México, México
³Laboratorio de Control Metabólico, Carrera de Medicina, Facultad de Estudios Superiores Iztacala, Universidad Nacional Autónoma de México, Tlalnepantla de Baz, Estado de México, México
*Corresponding author: Rodríguez-Enríquez S Laboratorio de Control Metabólico, Carrera de Medicina, Facultad de Estudios Superiores Iztacala, Universidad Nacional Autónoma de México, México. Email: saren960104@hotmail.com; sara.rodriguez@iztacala.unam.mx; saren960104@hotmail.com
Received: March 08, 2023 Accepted: April 22, 2023 Published: April 29, 2023
Abstract
The advent of natural phytochemicals for interfering with the HPI/AMF (erythrose, Ery) and TGF-ß (sulforaphane, SP) signaling pathways appears as a promising therapeutic strategy against Breast Cancer Stem Cells (BCSC). The BCSC viability, drug resistance, apoptosis evasion, invasiveness and HPI/AMF signaling axe were analyzed with Ery or SP. HPI/AMF, its downstream targets (H-Ras, p-Akt, p-Erk, NF-kB, p-NF-kB, Wnt/ß-catenin), XIAP, cytokines secretion and the glycoprotein- P increased their contents (2-83 times) in BCSC vs. parental MCF-7 cells. The incubation of BCSC with Ery (80nM/24h) significantly decreased (>85%) the HPI/AMF level, glycoprotein-P, XIAP and the secretion of HPI-AMF/TGF-ß vs. non treated-BCSC, which in turn affected cellular invasiveness. SP (150μM/24h) was unable to affect HPI/AMF axe, although invasiveness was affected (>80%). Ery in combination with SP decreased BCSC viability after 3 days of treatment. These results showed that these phytochemicals are a promising nutrition strategy prompting the design of treatments to blocking cancer relapse induced by CSC.
Keywords: Breast cancer stem cells; Erythrose; Sulphoraphane; Metastasis; Drug resistance
Introduction
The growth of Cancer Stem Cells (CSC) involves the activation of several signaling pathways associated to cell surviving (i.e., Wnt/ß-catenin, Hedgehog, Notch, and BMI1) [1-4]. To overcome the observed CSC therapeutic resistance, some surviving pathway-targeted therapies have been recently proposed [3,5]. In this regard, SMO/Hedgehog (cyclopamine, GDC-0449, IPI-926, BMS-83392) and Notch (MK-0752) inhibitors have been tested in CSC from medulloblastoma and basal cell, pancreas and breast carcinomas [3,6]. However, high resistance and severe side effects (diarrhea, nausea, vomiting, and fatigue) in drug-treated patients was observed [3,7]. Therefore, effective anti-CSC strategies should focus on the identification of signaling pathways principally found in CSC but absent or not frequently found in non-cancer cells [8]. Breast Cancer Stem Cells (BCSC) actively secretes HPI/AMF (hexose phosphate isomerase/autocrine motility factor) from cytosol to extracellular milieu [9]. As cytokine, external HPI/AMF binds to its specific plasma membrane receptor Pg78, trig- gering several processes related with the CSC phenotype development [9,10]. Interestingly, Erythrose-4 Phosphate (E4P) at nanomolar doses (24nM) blocks the release of HPI/AMF from the cytosol to extra- cellular milieu, decreasing the content of stemness associated-proteins, mammospheres formation and tumor invasiveness in BCSC [11].
Although E4P treatment was therapeutically attractive for BCSC, it induced heart failure [12]. In addition, it is costly, which may limit its use in the clinical practice. Thus, to identify E4P analogues with high anti-cancer potential and better cost-benefit balance, D-Erythrose (Ery) was tested as a suitable anticancer phytochemical for BCSC treatment targeting the HPI/AMF signaling pathway. Ery is a root rhubarb phytochemical promoting apoptosis in mice colon [13] and LL-2 Lewis lung [14] carcinomas with low price and scarce or negligible side effects [13].
On the other hand, the phytochemical Sulforaphane (SP) derived from cruciferous vegetables has also shown anticancer effect on non-small cell lung cancer [15] and affects the prostate CSC development, diminishing the level of several proteins (NF- kB, Sox-2 and ALDH1) associated with stemness [16] as well as interleukins and TGF-ß [17].
In order to assess the effect of Ery or SP on the development of stem cells, an integral analysis was carried out by measuring cell viability, the HPI/AMF signaling axis, stemness protein levels, and EMT and cellular invasion processes in the presence of Ery or SP in human breast MCF-7 CSC. In parallel and as a control, the effect of Ery or SP on proliferation and cellular viability of non-cancer cells (human HUVEC and mouse 3T3 fibro- blasts) was also analyzed. Our present study may help to elucidate potential natural nutrition-based treatments for BCSC treatment and elimination.
Materials and Methods
Cell Culture
Human breast low-metastatic MCF-7 cells, human breast metastatic MDA-MB-231 cells and mouse fibroblasts 3T3 (1x106 cells/mL) were grown in Dulbecco-MEM (DMEM). Human umbilical vein endothelial cells or HUVEC (1x106 cells/mL) were grown in Eagle's MEM. All culture media were supplemented with 10% fetal bovine serum (GIBCO; Rockville, USA) plus 10,000 U penicillin/streptomycin (Sigma; Steinheim, Germany). Cells were cultured in a humidified atmosphere of 5% CO2/95% air at 37°C for 3-4 days until confluence of 80-90% was reached. Genotyping of the MCF-7 and MDA-MB-231 cell lines used in the present work at the National Institute of Genomic Medicine, Mexico revealed that both cell lines shared all allelic markers (14 out of 14) with their original clones. HUVEC were kindly provided by Dr. R. López-Marure from Departamento de Fisiología at Instituto Nacional de Cardiologia, México.
Breast Cancer Stem Cells (BCSC) Selection and Isolation
BCSC were isolated from MCF-7 and MDA-MB-231 parental cell lines following the stem cell isolation protocol previously reported [11]. Briefly, MCF-7 cells (1X106 cells/mL) were exposed to hypoglycemia (2.5mM glucose) plus taxol (100nM) for 12h. Afterwards, old medium was replaced by complete DMEM containing 25mM glucose plus doxorubicin (100nM) and further exposed to severe hypoxia (0.1% O2) for additional 12h inside an Oxygen Control Chamber (Coy Laboratory, Grass Lake, MI). Finally, cells were cultured in serum-free DMEM containing 25mM glucose and placed in a humidified atmosphere of 5% CO2/95% air at 37°C for 24h, until their use [11].
Western Blot and Immunoprecipitation Assays
BCSC and MCF-7 (5x106 cells/mL) cells were dissolved in RIPA lysis buffer containing Phosphate Buffer Saline (154mM NaCl, 5mM Na2HPO4 and 1.5mM KH2PO4) 1xpH 7.2, 1% IGEPAL NP40, 0.1% SDS and 0.05% sodium deoxycholate. RIPA was complemented with 1mM PMSF (phenyl methanesulfonyl fluoride) and 1 tablet of complete protease inhibitors cocktail (Roche, Mannheimm, Germany) [9]. Protein samples (40μg) resuspended in loading buffer containing 4M glycerol, 0.05% bromophenol blue, 10% SDS and 0.5M Tris-Cl/SDS, pH 6.8 plus 5% ß-mercaptoethanol were loaded onto 10 or 12.5% polyacrylamide gel under denaturalizing conditions. Afterwards, electrophoretic transfer to PVDF membranes (BioRad; Hercules, CA, USA) was performed followed by overnight immunoblotting with different antibodies. 1:1000 dilutions of CD44, ALDH1A3, Oct3/4, HPI/AMF, NFkB/p65, p-NF-KB p65 (S536), XIAP, Wnt, ß-catenin antibodies; and 1:500 dilution of HPI/AMF receptor (gp78), H-Ras, Akt, p-Akt (S473), Erk1/2, p-Erk1/2, p-glycoprotein, IL-8, IL-6, TGF-ß and a-tubulin antibodies (Santa Cruz; Santa Cruz, CA, USA) were used at 4°C. The corresponding secondary antibodies conjugated with horseradish peroxidase (Santa Cruz Biotechnology) revealed the hybridization bands. The signal was detected by chemiluminescence using the ECL-Plus detection system (Amersham Bioscience; Little Chalfont, Buckinghamshire, UK). Densitometry analysis was performed using the Scion Image Software (Scion; Bethesda MD, USA) and normalized against its respective load control. Percentage of each band represents the mean±S.D. of at least three experiments carried out with independent biological samples.
To reveal HPI/AMF-gp78 complex, immunoprecipitation assays were performed as follows. Proteins (1mg/mL) were immunoprecipitated by incubating the total cellular protein content with the HPI/AMF antibody (5μg/mL) for 1h plus protein A-sepharose (Sigma, Steinheim, Germany). After boiling for 3-5 min, eluted proteins were loaded onto 10 or 12.5% polyacrylamide gel under denaturalizing conditions. Afterwards, electrophoretic transfer to PVDF membranes was performed, followed by overnight immunoblotting with anti-HPI/AMF and anti-gp78 antibodies and by using their respective secondary antibodies following manufacturer instructions. IgG1 (5μg/mL) was used for loading control. Densitometry analysis was performed by using the Scion Image Software (Scion, Bethesda MD, USA).
Spheroids Growth Assay
BCSC spheroids were generated by using the floating sphere-forming assay [18]. Briefly, BCSC, drug-treated BCSC and original MCF-7 cells (1X105 cells) were grown in Erlenmeyer flasks with free serum-DMEM and placed immediately under slow (20-50 rpm) orbital shaking for 10 days at 37°C in 95% air/5% CO2. Fresh DMEM was added every 2-3 days to replace used medium and remove cellular debris and planktonic cells not-forming spheroids. Spheroids size was measured at different culture times with a graduated reticule in an inverted phase contrast microscope (1/10mm; Zeiss, NY, USA).
Detection of HPI/AMF Protein in the Extracellular Milieu of BCSC
Extracellular Hexose Phosphate Isomerase (HPI) protein detection was assayed in cell-free DMEM from 24 h BCSC and MCF-7 cell cultures. Cell-free medium was incubated with 10% trichloroacetic acid at 4°C overnight [9]. Afterwards, the mixture was centrifuged once at 10,000rpm for 30 min and 4°C. The sediment was resuspended in loading buffer and loaded onto 12.5% SDS-PAGE gels. Electrophoretic transfer to PVDF membranes was followed by overnight immunoblotting with 1:500 dilution of HPI/AMF antibodies (Santa Cruz, CA USA) at 4°C. The hybridization bands were revealed with the mouse secondary antibody conjugated with horseradish peroxidase (Santa Cruz, CA USA). The signal was detected by chemiluminescence as described above. Albumin was used as load control.
Invasiveness Assays
BCSC (5x104 cells/mL) were resuspended in serum-free DMEM and placed in the upper compartment of Boyden chambers (Trevigen Inc., Helgerman, USA) for 24h at 37°C. The lower compartment of the chamber was filled with serum-free DMEM. Calcein acetomethylester (calcein-AM, 60nM) (Trevigen Inc., Helgerman, USA) was added to the lower Boyden compartment for cell detection after 60 min incubation at 37°C, and after 24h of seeding cells in the upper compartment. Calcein fluorescence was detected at 485nm excitation and 520nm emission in a microplate reader (NunclonTM, Roskilde, Denmark). Metastatic breast cancer MDA-MB-231 cells were used to set up the maximal (100%) invasiveness capacity [9].
Drugs Treatment
BCSC (1x105 cells/0.5mL) were seeded in 24-well plates in DMEM for 24h. Afterwards, Erytrose (Ery) was added at final concentrations of 0.1, 1, 10, 100 nM; 1, 10, 100 μM and 1 or 10 mM for additional 24h. In another parallel set of experiments, Sulforaphane (SP) was added at final concentrations of 0.1, 1, 10, 100 nM; 1, 10, 100 μM and 1 or 10 mM for additional 24h. For comparative purposes, canonical anti-breast cancer drugs Doxorubicin (DOXO) and Paclitaxel (PA) were also assayed at the same concentrations used for Ery. At the end of each cellular culture (24h), viability was determined by trypan blue stain assay [19]. The IC50 value (i.e., concentration required to decrease cellular viability by 50%) was calculated by using the Origin 8 software (Northampton MA, USA) [20].
Glycoprotein P Activity
This activity was evaluated by using a microplate reader (NunclonTM, Roskilde, Denmark) in the presence of 0.25μM calcein-AM. Dye uptake was measured after 30 min incubation at 37°C using 485nm excitation and 520nm emission [21].
Statistical Analysis
Data shown represent the mean±Standard Deviation of the indicated number of independent experiments. The experimental and control groups were statistically compared using ANOVA and post-hoc Scheffé tests with P values <0.01 or <0.05 as significance criterion [22,23].
Results
Breast Cancer Stem Cell Phenotype
BCSC were isolated and enriched by a selective sequential method using hypoxia, hypoglycemia and drug exposure [11]. BCSC derived from MCF-7 cells showed high viability (>85%) and the enriched presence of classical stemness phenotype markers as described below. (i) Over-expression by 9.5-81 times of the typical stemness and pluripotency protein markers CD44+, ALDH1A3 and Oct-3/4 (Figure 1A). (ii) Overexpression by 88.5 times of the intracellular HPI/AMF receptor gp-78 and cytosolic HPI/AMF (Figure 1A). (iii) The acquisition of invasive phenotype associated to Epithelial-Mesenchymal Transition (EMT) (Figure 1B). (iv) The ability to secrete high levels of cytosolic HPI/AMF to the extracellular milieu (Figure 2) which in turn, binds to its highly expressed gp78 receptor (Figure 1A). (v) Typical fibroblastoid stem cell morphology (data not shown). (vi) Spheroids formation in free-serum medium (Figure S1). (vii) Acquisition of drug resistance towards Paclitaxel (PA) and Doxorubicin (DOXO) (Figure S2).
BCSC also showed increased levels (8-83 times) of several HPI/AMF-downstream signaling proteins such as H-Ras, p-NF-kB, Wnt/ß-catenin and the phosphorylated isoforms p-Akt and p-Erk; as well as a significant increment in the anti-apoptotic protein XIAP and in the protein related with drug resistance P-glycoprotein (Figure 1). The higher protein level of BCSC P-glycoprotein correlated with its enhanced activity [24] (Figure S3). The data on P-glycoprotein activity indicated that the rate of the fluorescent calcein uptake in BCSC was significantly lower (58%) than that attained in the original MCF-7 cells, suggesting more active calcein extrusion by the multidrug P-glycoprotein transporter (Figure S3). BCSC developed a greater invasiveness (40-times) compared to their MCF-7 parental cell line and similar to that displayed by the well-known highly malignant and invasive triple negative breast cancer MDA-MB-231 cells (Figure 3).
Effect of Metabolic Inhibitors and Canonical Anticancer Drugs on MCF-7 BCSC Viability
The values of IC50 on MCF-7 derived BCSC viability for Ery, SP and the canonical cancer drugs Doxorubicin (DOXO) and Paclitaxel (PA) are shown in Table 1. For comparison, the effect of all these drugs was tested (1) in other breast CSC isolated from human triple negative breast MDA-MB-231 cells and (2) non-cancer cells. The MDA-MB-231 BCSC were obtained by using the BCSC isolation protocol previously reported [11], and which revealed a significant increase (8-times vs. parental cell line) in their canonical stemness marker CD44+ (data not shown).
Drugs
Parental
Cancer Stem Cells
Non-Cancer Cells
MCF-7
MDA-MB-231
MCF-7
MDA-MB-231
HUVEC
3T3
Ery
25±2μM
32±3μM
81±4nM
114(2)nM
>)(Mμ01
>5mM(2)
SP
5.7±0.6μM
35±2.5μM
150±7.5nM
134±4nM
>)(Mμ01
>5mM(2)
DOXO
7±0.5μM
7±1.5μM
23±2μM
155±6μM
0.1mM (2)
0.36mM (2)
PA
5±0.4μM
22±4μM
75±21μM
98±3μM
> 5mM (2)
2.3mM (2)
Abbreviations: Ery: Erythrose; SP: Sulphoraphane; DOXO: Doxorubicin; PA: Paclitaxel
Table 1: Drug viability IC50 values in cancer and non-cancer cells. Drug treatment protocol and IC50 viability value determination are indicated in Methods section. Data shown represents at least 3 independent experiments, except where otherwise indicated in parenthesis.
After 24h exposure, viability IC50 values in BCSC from both MCF-7 and MDA-MB-231 cells were in the nanomolar range for Ery and SP, whereas their respective IC50 values in the parental cell lines were in the micromolar range. In contrast, BCSC IC50 values were in the micromolar range for DOXO and PA, but remarkably these values were much higher (>3 times) than those in their parental cell lines.
In order to establish the drug toxicity in non-cancer cells, the viability IC50 values of the tested drugs were also determined in HUVEC and 3T3 fibroblasts (Table 1). Much greater doses (>5mM) of Ery or SP were required to decrease by 50% HUVEC or 3T3 cell viability, indicating that these phytochemicals may effectively and selectively kill BCSC, but they do not have any apparent effect on normal cells. PA was also more effective for decreasing viability of BCSC and parental cancer cells than that of non-cancer cells (Table 1). On the contrary, DOXO showed a severe toxic effect on HUVEC and 3T3 cells at concentrations at which BCSC viability was also compromised (Table 1), illustrating the typical unspecific effect of DOXO [25]. The viability IC50 value of Ery (80nM) and SP (150nM) calculated for MCF-7 BCSC (Table 1) was used in the following set of experiments in which the effect of a single phytochemical or drug was analyzed.
Erythrose (Ery) Abolishes HPI/AMF Signaling, as well as Viability and Invasiveness Potential in MCF-7 BCSC
The exposure to Ery (80nM/24h) decreased some functional features associated to the BCSC phenotype. For instance, (1) the content of several stemness markers such as CD44+, Oct 3/4 and ALDH1 (Figure 1A); (2) the ability for spheroids formation in free-serum medium (Figure 1); and (3) the cell invasiveness potential (Figure 3) were significantly depressed by more than 90% vs. non-treated BCSC. In addition, proteins related with apoptosis regulation (XIAP), epithelial-mesenchymal transition (Wnt-1, β-catenin) and drug resistance (P-gp) also decreased by 70-95% after 24h incubation with the phytochemical (Figure 1). The HPI/AMF signaling axe was particularly targeted. Ery diminished the content (Figure 1A) and activity (Figure S3) by 84-97.5% of the HPI/AMF receptor (gp78) and some signaling downstream HPI/AMF proteins such as H-Ras, p-Akt, and p-Erk1/2 (Figure 1A).
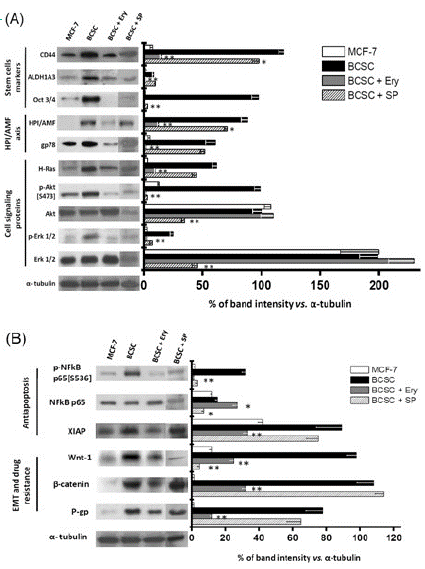
Figure 1: Effect of Ery (80nM/24h) or SP (150nM/24h) on proteins related with (A) stemness and HPI/AMF axes; and (B) Apoptosis, EMT and drug resistance. Data shown rep-resents at least three different preparations. *P<0.05 and **P<0.01 vs. BCSC.
To establish whether external Ery impairs the HPI/AMF cytokine function by blocking the HPI/AMF-gp78 interaction, co-immunoprecipitation assays were carried out. The gp78 antibody indeed recognized the formation of the HPI/AMF-gp78 complex (Figure 2B), as previously described [11]. However, this complex was not apparent in the presence of 80nM external Ery (Figure 2B).
After stemness phenotype induction, HPI/AMF, interleukins (IL-6, IL-8) and the transforming growth factor (TGF-β) are secreted to the extracellular medium for metastasis onset [26]. Indeed, higher contents of HPI/AMF (186-times), TGF-β (3-times) and interleukins 6 and 8 (2.5-4 times) were detected in the extracellular BCSC medium compared to that from parental MCF-7 cells (Figure 2A). After Ery (80nM/24h) treatment, the secretion of HPI/AMF and IL-6 was severely blocked; whereas IL-8 and TGF-β secretion was unaffected by the drug, indicating that Ery may specifically block HPI/AMF secretion without affecting other interleukin and cytokine secretion, except for that of IL-6 and TGF-β.
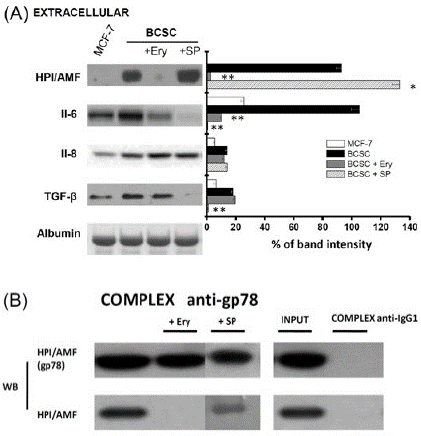
Figure 2: Effect of Ery (80nM/24h) or SP (150nM/24h) on (A) extracellular HPI/AMF, interleukins and TGF-β; and (B) Immunoprecipitation of BCSC HPI/AMF and gp78. Data shown represents at least three different preparations. *P<0.05 and **P<0.01 vs. BCSC.
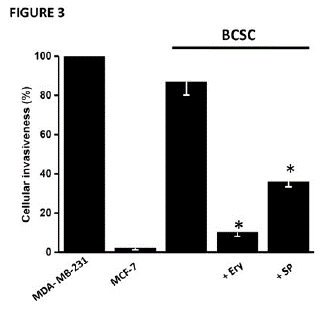
Figure 3: Effect of Ery (80nM/24h) or SP (150nM/24h) on BCSC cellular invasiveness. Data shown represents at least three different preparations. *P<0.01 vs. BCSC.
Contrary to what was observed with erythrose, SP (150nM/24h) was not able to strongly block the expression of proteins associated with stemness (CD44+ and ALDH1A3), HPI/AMF axe (gp78 receptor, cytosolic HPI/AMF, H-Ras), anti-apoptosis (XIAP), EMT (β-catenin) and drug resistance (level and activity of P-gp) (Figure 1). In addition, SP did not block the formation of the gp78-HPI/AMF complex nor the secretion of HPI/AMF cytokine, although the release of interleukins and TGF-β was severely compromised (Figure 2A) as previously reported [17,27,28]. In spite of HPI/AMF axe was not affected, the BCSC invasiveness ability was greatly affected by SP, indicating that SP has targets other than the HPI/AMF axe for avoiding invasiveness.
Effect of Ery Plus SP on MCF-7 BCSC Viability
Several reports indicate that SP has a synergic effect on chemotherapy-based drugs like cisplatin, PA or DOXO [29]. In order to assess whether SP (150nM) indeed increases toxicity of Ery (80nM), PA (98μM) or DOXO (155μM), the combination of these drugs was tested on the viability of BCSC and compared to that of non-cancer cells (Figure 4).
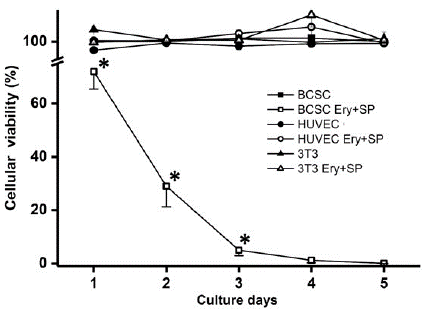
Figure 4: Effect of Ery (80nM) plus SP (150nM) on BCSC cellular viability. Data shown represents at least three different preparations. *P<0.05 vs. BCSC.
Viability decreased by 28% from day 1 of culture when SP + Ery were added simultaneously to the BCSC culture. At days 2 and 3 of culture, viability was significantly decreased by 71 and 95%, respectively, whereas viability of non-cancer cells was unaffected (Figure 4).
Discussion
The key inducing factors involved in the development of the triple negative MDA-MB-231 BCSC are hypoxia, hypoglycemia and drug exposure [11]. This multi-stress cell selection protocol appeared to be useful for CSC enrichment, independently of cancer cell type. Hypoxia stabilizes HIF-1a, which in turn, promotes the over-expression of several stemness proteins particularly in breast cancer [30,31]. Hypoglycemia activates kinases (AMPK) associated with stem cell growth, self-renewal and differentiation [30,31]. It is noted that in the present study, drug resistance was also evaluated in both MCF-7 and MDA-MB-231, since it represents the principal factor to be considered in the therapy against CSC because it is linked to high recurrence, poor prognosis, poor clinical outcome, progression and death [32,33].
HPI/AMF Axe as Molecular Target to Induce BCSC Death
The HPI/AMF surviving signaling pathway and some of their down-stream proteins (H-Ras, p-Akt, p-Erk, NF-kB and Wnt/β-catenin) have been widely studied in cancer cells but not specifically in cancer stem cells. This takes relevance because CSC maintain distinguishing characteristics regarding surviving pathways vs. the rest of cancer cells [34,35]. Indeed, BCSC show increased levels of HPI/AMF protein content and its extracellular receptor gp-78 (present study) [11]. Interestingly, these increments in HPI/AMF/gp-78 levels in BCSC correlated with (1) a significant elevation in the levels of p-Akt, p-Erk, NF-kB and Wnt/β-catenin (i.e., Figure 1); and (2) increments in pathway functionality measured as high levels in EMT proteins and invasiveness (i.e., Figure 3). This positive relationship between HPI/AMF signaling axe, EMT and invasiveness has been also observed in breast and brain metastatic cancer cells [36,37], which have a mixed population of CSC and non-CSC.
These observations suggest that HPI/AMF is a potential promising therapeutic target for CSC because (i) this surviving pathway is not predominantly found in non-cancer cells [38,39]; and (ii) some inhibitors directed against HPI like E4P seem to be innocuous in mouse fibroblasts [9]. Although E4P blocks the cytokine role of HPI/AMF deterring BCSC invasiveness [9] and promotes apoptosis in several carcinoma (breast, brain, pancreas, colorectal) cells [14], the doses used caused glucose-6-phosphate accumulation in myocytes, which in turn, activated mTOR pathways causing rat heart failure [12]. Thus, E4P seemed not to be a suitable drug candidate for cancer treatment.
Therefore, in the search for HPI inhibitors with negligible effects on non-cancer cells [13], Erythrose (Ery) emerged as a potential alternative for BCSC treatment.
Ery as Therapeutic Drug against BCSC
Ery is a phytochemical contained in rhubarb roots with apparent anticancer effects diminishing the size of intraperitoneal metastatic colon carcinomas in mice [13] and subcutaneous LL-2 Lewis lung carcinoma in mice [14].
It has been established that one of the molecular mechanisms associated to Ery effect in cancer cells is its ability to become transformed into carbonic acid by carbonic anhydrase [14], thus promoting cellular death induced by acidosis [13]. In our study, the ability of Ery to act as a multi-target inhibitor by blocking the HPI/AMF axe was also shown. This observation may be relevant because the stemness process is highly dependent on specific cytokines that regulate malignant progression [40]. In this regard, in the present study it was shown that Ery specifically attenuated CSC malignancy by 1) blocking the release of HPI/AMF from cytosol to extracellular milieu, similarly to E4P [9]; 2) inhibiting the gp-78/ HPI/AMF interaction; and consequently 3) inactivating the HPI/AMF down-stream signaling pathway.
In contrast to other anticancer approaches using antibodies such as adalimumab (8μg/mL), whose targets are extracellular interleukins and TGF-β, which may compromise non-cancer cell functionality [41]. Ery did not affect interleukins and TGF-β secretion, suggesting it’s selectively role as HPI/AMF blocker.
Sulforaphane (SP) was certainly able to block interleukins and TGF-β secretion by BCSC (c.f. Figure 2A), as it was reported for neuroblastoma and hepatocarcinoma cells [42,43]. However, SP did not affect the level of proteins related with the HPI/AMF axe (including HPI/AMF/gp78 interaction and HPI/AMF secretion). These last results clearly indicated that other surviving signaling pathways, instead of the HPI/AMF axe, might have mediated the SP inhibition of stem cell invasiveness and viability. SP downregulates c-MYC and β-catenin levels affecting stemness properties associated to miR-214 in non-small stem cell lung cancer [15]. SP also diminishes the transcription factor NF-kB, Sox-2 and ALDH-1 activity; it eliminates self-renewal potential in prostate cancer stem-like cells [16]; and it improves chemotherapy in gastric cancer by activating interleukin-6/interleukin receptor [44].
Ery and SP Combination Eliminates BCSC
Several attempts to kill CSC have been carried out by using different drugs or natural compounds [45-47]. At doses assayed in the present study (i.e., the determined IC50 viability values), the HPI/AMF axe was compromised impacting the metastasis process but keeping high BCSC viability. This finding concurs with clinical observations where several antineoplastic drugs (vemurafenib, nilotinib, sorafenib, imatinib) targeting specific BCSC signaling pathways are able to decrease metastasis at the beginning of treatment. However, several months after clinical treatment, cancer recurrence is observed [48,49].
Encouragingly, the effect of combining Ery plus SP on viability of BCSC was fully effective. Combinations of both phytochemicals were innocuous on non-cancer HUVEC and 3T3 mouse fibroblasts, providing experimental evidence for their use as a potential promising clinical strategy for BCSC treatment.
Conclusion
The use of phytochemicals like Ery to block BCSC activation of survival pathways emerge as a potential strategy for BCSC elimination. Ery was able to potently inhibit HPI/AMF pathway activation and in consequence, the development of the stemness phenotype i.e., decreased BCSC epithelial to mesenchymal transition, as well as decreased invasiveness and viability with no apparent effect on non-cancer cells. Therefore, future research might be focused on the use of these natural compounds, in combination with canonical drugs, to block the cancer stemness development and specially promote BCSC elimination.
Author Statements
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any potential conflict of interest.
Acknowledgements
The authors also grateful thank Farmaconsa S.A. de C. V. for its support in facilitating the writing and editing of the present manuscript, and the design of some experiments.
Funding
The present work was partially supported by grants from CONACyT-México to SRE (No. 283144), SCPV (No. 377873) and RMS (No. 6379); and No. IA201823 (SR-E) from PAPIIT, DGAPA-UNAM
References
- Chappell W, Steelman L, Long JM, Kempf RC, Abrams SL, et al. Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR inhibitors: rationale and importance to inhibiting these pathways in human health. Oncotarget. 2011; 2: 135-164.
- Polakis P. Wnt signaling in cancer. Harb Perspect Biol. 2012; 4: a008052.
- Borah A, Raveendran S, Rochani A, Maekawa T, Kumar DS. Targeting self-renewal pathways in cancer stem cells: clinical implications for cancer therapy. Oncogenesis. 2015; 4: e177.
- Jang GB, Kim JY, Cho SD, Park KS, Jung JY, et al. Blockade of Wnt/β-catenin signaling suppresses breast cancer metastasis by inhibiting CSC-like phenotype. Sci Rep. 2015; 5: 12465.
- Fang L, Zhu Q, Neuenschwander M, Specker E, Wulf-Goldenberg A, et al. A small-molecule antagonist of the β-Catenin/TCF4 interaction blocks the self-renewal of cancer stem cells and suppresses tumorigenesis. Cancer Res. 2016; 76: 891-901.
- Schott AF, Landis MD, Dontu G, Griffith KA, Layman Rm, et al. Preclinical and clinical studies of gamma secretase inhibitors with docetaxel on human breast tumors. Clin Cancer Res. 2013; 19: 1512-1524.
- Yuan X, Wu H, Xu H, Xiong H, Chu Q, et al. Notch signaling: an emerging therapeutic target for cancer treatment. Cancer Lett. 2015; 369: 20-27.
- Dodge ME, Lum L. Drugging the cancer stem cell compartment: lessons learned from the hedgehog and Wnt signal transduction pathways. Annu Rev Pharmacol Toxicol. 2011; 51: 289-310.
- Gallardo-Pérez JC, Rivero-Segura NA, Marín-Hernández A, Moreno-Sánchez R, Rodríguez-Enríquez S. GPI/AMF inhibition blocks the development of the metastatic phenotype of mature multi-cellular tumor spheroids. Biochim Biophys Acta. 2014; 1843: 1043-1053.
- Wiseman E, Zamuner A, Tang Z, Rogers J, Munir S, et al. Integrated multiparametric high-content profiling of endothelial cells. SLAS Discov. 2019; 24: 264-273.
- Gallardo-Pérez JC, Adán-Ladrón de Guevara A, Marín-Hernández A, Moreno-Sánchez R, Rodríguez-Enríquez S. HPI/AMF inhibition halts the development of the aggressive phenotype of breast cancer stem cells. Biochim Biophys Acta. 2017; 1864: 1679-1690.
- Karlstaedt A, Khanna R, Thangam M, Taegtmeyer H. Glucose 6- phosphate accumulates via phosphoglucose isomerase inhibition in heart muscle. Circ Res. 2020; 126: 60-74.
- Liu LL, Yi T, Zhao X. Antitumor effect of D-erythrose in an abdominal metastatic model of colon carcinoma. Oncol Lett. 2015; 9: 769-773.
- Wang X, Wei Y. Erythrose kills cancer cell in vitro and inhibit tumor growth in vivo. Cancer Res. 2010; 70: 4548.
- Li QQ, Xie YK, Wu Y, Li LL, Li Y, et al. Sulforaphane inhibits cancer stem-like cell properties and cisplatin resistance through miR-214-mediate downregulation of c-MYC in non-small cell lung cancer. Oncotarget. 2017; 8: 12067-12080.
- Labsch S, Liu L, Bauer N, Zhang Y, Aleksandrowicz E, et al. Sulforaphane and TRAIL induce a synergistic elimination of advanced prostate cancer stem-like cells. Int J Oncol. 2014; 44: 1470-1480.
- Liang J, Jahraus B, Balta E, Ziegler JD, Hübner K, et al. Sulforaphane Inhibits Inflammatory Responses of Primary Human T-Cells by Increasing ROS and Depleting Glutathione. Front Immunol. 2018; 9: 2584.
- Manuel-Iglesias J, Beloqui I, Garcia-Garcia F, Leis O, Vazquez-Martin A, et al. Mammosphere formation in breast carcinoma cell lines depends upon expression of E-cadherin. PLoS One. 2013; 8: e77281.
- Rodríguez-Enríquez S, Pacheco-Velázquez SC, Marín-Hernández A, Gallardo-Perez JC, Robledo-Cadena DX, et al. Resveratrol inhibits cancer cell proliferation by impairing oxidative phosphorylation and inducing oxidative stress. Toxicol Appl Pharmacol. 2019; 370: 65-77.
- Greco WR, Bravo G, Parsons JC. The search for synergy: a critical review from a response surface perspective. Pharmacol Rev. 1995; 47: 331-385.
- Holló Z, Homolya L, Davis CW, Sarkadi B. Calcein accumulation as a fluorometric functional assay of the multidrug transporter. Biochim Biophys Acta. 1994; 1191: 384-388.
- Klockars AJ, Hancock GR. Scheffe´s more powerful F-protected post hoc procedure. J Educ Behav Stat. 2000; 25: 13-19.
- Krzywinski M, Altman N. Points of significance: Comparing samples-part I. Nat Methods. 2014; 11: 215-216.
- Sharom J. ABC multidrug transporters: structure, function and role in chemoresistance. Pharmacogenomics 2008; 9: 105-127.
- Thorn CF, Oshiro C, Marsh S, Hernandez-Boussard T, McLeod H, et al. Doxorrubicin pathways: pharmacodynamics and adverse effects. Pharmacogenet Genomics. 2011; 21: 440-446.
- Wu Y, Wang Y, Lin Y, Liu Y, Wang Y, et al. Dub 3 inhibition suppresses breast cancer invasion and metastasis by promoting Snail1 degradation. Nat Commun. 2017; 8: 14228.
- Shankar S, Ganapathy S, Srivastava RK. Sulforaphane enhances the therapeutic potential of TRAIL in prostate cancer orthotopic model through regulation of apoptosis, metastasis, and angiogenesis. Clin Cancer Res. 2008; 14: 6855-6866.
- Townsend BE, Johnson RW. Sulforaphane reduces lipopolysaccharide-induced proinflammatory markers in hippocampus and liver but does not improve sickness behavior. Nutr Neurosci. 2017; 20: 195-202.
- Kamal MM, Akter S, Lin CN, Nazzal S. Sulforaphane as an anticancer molecule: mechanism of action, synergistic effects, enhancement of drug safety, and delivery systems. Arch Pharm Res. 2020; 43: 371-384.
- Conley SJ, Gheordunescu E, Kakarala P, Newman B, Korkaya H, et al. Antiangiogenic agents increase breast cancer stem cells via the generation of tumor hypoxia. Proc Natl Acad Sci USA. 2012; 109: 2784-2789.
- Xie J, Xiao Y, Zhu XY, Ning ZY, Xu HF, Wu HM. Hypoxia regulates stemness of breast cancer MDA-MB-231 cells. Med Oncol. 2016; 33: 42.
- Bertolini G, D´Amico L, Moro M, Landoni E, Perego P, et al. Microenvironment-Modulated metastatic CD133+/CXCR4+/EPCAM-lung cancer-initiating cell sustain tumor dissemination and correlate with poor prognosis. Cancer Res. 2015; 75: 3636-3649.
- Huang J, Li H, Ren G. Epithelial – mesenchymal transition and drug resistance in breast cancer. Int J Oncol. 2015; 47: 840-848.
- Gong S, Li Q, Jeter CR, Fan Q, Tang DG, et al. Regulation of NANOG in cancer cells. Mol Carcinog. 2015; 54: 479-487.
- De Sousa EMF, Vermeuen L. Wnt signaling in cancer stem cell biology. Cancers. 2016; 8: 60.
- Kho DH, Zhang T, Balan V, Wang Y, Ha SW, et al. Autocrine motility factor modulates EGF-mediated invasion signaling. Cancer Res. 2014; 74: 2229-2237.
- Li Y, Wei Z, Dong B, Lian Z, Xu Y. Silencin of phosphoglucose isomerase/autocrine motility factor decreases U87 human glioblastoma cell migration. Int J Mol Med. 2016; 37: 998-1004.
- Maruyama K, Watanabe H, Shiozaki H. Expression of autocrine motitlity factor receptor in human esophageal squamus cell carcinoma. Int J Cancer. 1995; 64: 316-321.
- Takanami I, Takeuchi K. Autocrine motitily factor-receptor gene expression in lung cancer. Jpn Thorac Cardiovas Surg. 2003; 51: 368-373.
- Korkaya H, Liu S, Wicha MS. Regulation of cancer stem cells by cytokine networks: attacking cancer’s inflammatory roots. Clin Cancer Res. 2011; 17: 6125-6129.
- Grabarek BO, Wcislo-Dziadecka D, Sanakiewicz A, Kruszniewska-Rajs C, Gola J. Evaluation of changes in expression pattern of oxidative stress genes under the influence of adalimumab. Dermatol Ther. 2019; 32: e13141.
- Negi G, Kumar A, Sharma S. Nrf2 and NF-κB modulation by sulforaphane counteracts multiple manifestations of diabetic neuropathy in rats and high glucose-induced changes. Curr Neurovasc Res. 2011; 8: 294-304.
- Wu J, Han J, Hou B, Deng C, Wu H, Shen L. Sulforaphane inhibits TGF-β-induced epithelial-mesenchymal transition of hepatocellular carcinoma cells via the reactive oxygen species-dependent pathway. Oncol Rep. 2016; 35: 2977-2983.
- Wang X, Li Y, Dai Y, Liu Q, Ning S, et al. Sulforaphane improves chemotherapy efficacy by targeting cancer stem cell-like properties via the miR-124/IL-6R/STAT3 axis. Sci Rep. 2016; 6: 36796.
- Mann J. Natural products in cancer chemotherapy: past, present and future. Nat Rev Cancer. 2002; 2: 143-148.
- Nobili S, Lippi D, Witort E, Donnini M, Bausi L, et al. Natural compounds for cancer treatment and prevention. Pharmacol Res. 2009; 59: 365-378.
- Hoelder S, Clarke PA, Workman P. Discovery of small molecule cancer drugs: successes, challenges and opportunities. Mol Oncol. 2012; 6: 155-76.
- Italiano A, Cioffi A, Coco P, Maki RG, Schoffski P, et al. Patterns of care, prognosis, and survival in patients with metastatic gastrointestinal stromal tumors (GIST) refractory to first-line imatinib and second-line sunitinib. Ann Surg Oncol. 2012; 19: 1551-1559.
- Grimaldi AM, Simeone E, Ascierto PA. Vemurafenib plus cobimetinib in the treatment of mutated metastatic melanoma: the CoBRIM trial. Melanoma Manag. 2015; 2: 209-215.