Abstract
Fifty-four patients with acute myocardial infarction (MI) were examined using computer-assisted video biomicroscopy (CAVBM) of the bulbar conjunctiva. The patients were divided into three groups. Group 1 comprised 15 former smokers (mean age 54.3 ± 2.1). The mean duration of smoking in this group was 23.7 ± 2.4 years, and the mean time since smoking cessation was 11.8 ± 2 years. Group 2 was made up of 28 current smokers (mean age 51.8 ± 1.4). Group 3 consisted of 11 subjects who had never smoked (mean age 59.2 ± 1.5). Data collected from the former smokers were compared with data collected from the current smokers and those who had never smoked. In the former smokers, the mean arteriolar diameter, mean capillary diameter, arteriole-to-venule ratio, and number of capillaries per 1mm2 of conjunctiva were significantly higher than in the current smokers, but did not differ from the group of those who had never smoked. Thus, smoking adversely affects the microcirculatory bed (MCB) structure, which can recover after the cessation of smoking. The positive changes in the MCB of former smokers with ischemic heart disease (IHD) support the recommendation that patients with this condition stop smoking.
Keywords: Microcirculatory Bed; Myocardial Infarction; Smoking Cessation; Quitting Smoking
Introduction
Smoking is a modifiable risk factor for the development and progression of cardiovascular diseases; stopping smoking is the most effective way to prevent cardiovascular disorders and the premature deaths they caused. Despite smoking’s well–known negative effects on the development and progression of cardiac and vascular disorders, many patients continue to smoke even after a myocardial infarction. According to data published in 2017, 45% of patients being discharged from hospital after acute myocardial infarction (MI) continue to smoke, and only 10% of MI patients say at discharge that they “used to smoke but quit” [1]. It is also the case that the microcirculatory bed is the segment of the vascular system that is most susceptible to various factors, including exposure to nicotine in tobacco smoke [2-8]. In the 1980s the Department of Faculty Therapy at the Far Eastern State Medical University used biomicroscopy of the conjunctiva to study the effects of smoking on MCB in healthy young adults and young patients with ischemic heart disease (IHD) who had experienced an MI [7]. The results of the study, which compared young smokers with IHD and non-smokers, showed that tobacco smoking negatively affects microcirculation. Approaches to treating MI have changed; patients are now actively encouraged to stop smoking. Thus, comparing MCB parameters in MI patients who have stopped smoking, continue smoking, and have never smoked is now not only an academic issue, but also a practical one. Our study addresses the following question: Are smoking-induced MCB changes in MI patients reversible after these patients quit smoking (as they are in apparently healthy young people [2,3,6]), or do they remain unchanged?
Study Objective
To investigate the effects of smoking and the cessation of smoking on the microcirculatory bed in IHD patients with acute MI.
Materials and Methods
This study included 54 MI patients who had been hospitalized in the Cardiology or Myocardial Infarction Units of Khabarovsk Territorial Clinical Hospital No. 2. Using computer-assisted video biomicroscopy (CAVBM) of the bulbar conjunctiva, the microcirculatory bed was examined in all patients. This examination was done on day 7-10 of in-hospital treatment after each patient’s condition had stabilized. Microscopic photos of conjunctiva areas magnified 96 times were used to assess mean diameters of microvessels, arteriole-to-venule ratio, and number of capillaries per 1mm2 of conjunctiva. These measurements were taken with a microruler that had been calibrated before testing. All patients received treatment according to guidelines established for the management of MI in patients presenting with or without ST-segment elevation. Voluntary consent to participate in the study was obtained from all the patients. Study subjects were divided into three groups: Group 1 comprised 15 former smokers, aged 32 to 66 (the male/female ratio was 13:2, mean age 54.3 ± 2.1). Their duration of smoking varied from three to 35 years, with a mean duration of 23.7 ± 2.4 years; the time since smoking cessation ranged from eight months to 35 years, with the mean length of time11.8 ± 2 years (hereinafter M ± m, where M is the mean and m is the standard error of the mean). Group 2 was made up of 28 current smokers (the male/female ratio was 23:5, mean age 51.8 ± 1.4). Group 3 consisted of 11 patients who had never smoked (the male/female ratio was 3:8, mean age 59.2 ± 1.5). Data collected from former smokers were compared to data collected from current smokers and those who had never smoked. Exclusion criteria were: early MI complications (cardiogenic shock, heart rhythm disturbances, or pulmonary edema), a history of any heart-rhythm disturbances (atrial fibrillation, extra systoles, or atrio ventricular blocks), stage IIB-III chronic heart failure (CHF), inflammatory eye diseases, eye injuries, use of ophthalmic or nasal vasoconstrictors, concomitant inflammation of internal organs, systemic connectivetissue disease, asthma, chronic kidney disease stage 3 or higher, and neoplasms of any stage and location. Table 1 provides a detailed look at patient characteristics for all patient groups.
The data presented in Table 1 show that patients in all groups were matched by age, body mass index, hemodynamic parameters (blood pressure [BP] and pulse rate), and blood cholesterol and glucose levels. As in our previous studies, smoking was associated with the male gender. The intensity of smoking in groups 1 and 2 was similar. The duration of IHD in current smokers was minimal compared to that of former smokers, whose duration of IHD prior to MI was the longest (p1 = 0.0276).Group 1 subjects (former smokers) had higher rates of class II angina than subjects in groups 2 (current smokers) and 3 (those who had never smoked) (40.00%, 28.57%, and 18.18%, respectively). The percentage of patients without concomitant hypertension was 28.57% in group 2 (current smokers) compared to 6.67% in group 1 (former smokers) and 9.09% in group 3 (never smoked). The mean number of prior MI was similar in all groups. The study groups did not differ in the class of CHF. Q-wave MI was diagnosed in 80% of former smokers, 64.29% of smokers, and 63.64% of those who had never smoked (p1 = 0.2849 and p2 = 0.3527).
Statistical analysis was performed at the BIOSTATISTIKA Center, using statistical packages SAS 9.4 and STATISTICA 10. The critical-significance level for testing the null hypotheses was 0.05. Testing for normality of the distribution of quantitative parameters in the comparison groups was done using Kolmogorov-Smirnov, Shapiro-Wilk, Cramér–von Mises, and Anderson–Darling tests. Siegel–Tukey and Ansari-Bradley tests were used for checking the equality-of-dispersion hypotheses. As normal distribution was not shown for any of the analyzed parameters in any group, key study parameters were compared using the nonparametric Wilcoxon- Mann-Whitney test [4].
Contingency tables were used to analyze the association between pairs of categorical variables. In addition to analyzing the achieved level of statistical significance and the value of the chi-square test, the Cramér V-test was also used to assess the strength of the association between the study parameters [4].
Study Results
Table 2 shows the results of the comparisons of the group means that characterize changes in the caliber and number of microvessels (microcirculatory bed parameters) in the three study groups.
Table 2 data show that the former smokers had significantly higher mean arteriolar diameters than the current smokers (p1 < 0.0001). Thus, the arteriole-to-venule ratio in group 1 (former smokers) was also higher than in group 2 (current smokers) (p1 = 0.0003). Former smokers and those who had never smoked had similar mean arteriolar diameters (p2 = 0.2763). Venular parameters showed no statistically significant differences between study groups. Capillary diameters were significantly larger in former smokers than in current smokers (p1 = 0.0091). The lowest mean number of capillaries per 1mm2 of conjunctiva was seen in group 2 (current smokers) (2.3 ± 0.2units/ mm2), suggesting that tobacco smoking causes and further enhances rarification of capillaries in IHD patients. The mean number of capillaries per 1mm2 of conjunctiva in apparently healthy young people was 7.96 ± 0.25units/mm2 [2,6]. In group 1 (former smokers), the mean number of capillaries was significantly higher (4.7 ± 0.3; p1 ‹ 0.0001), which was similar to the values in group 3 (never smoked) (4.88 ± 0.3; p2 = 0.9392). Thus, although former smokers had MI, a severe IHD complication, rarification in these patients was less significant than in current smokers. This may be seen as a sign of capillary recovery.
Discussion of Results
CAVBM data of former smokers show that the microvasculature structure restores itself after the cessation of smoking, and this response does not depend on age, BMI, duration of IHD, depth of myocardial lesion, or laboratory data. In former smokers, MCB parameters were similar to those seen in patients who had never smoked. Thus, larger mean arteriolar and capillary diameters and higher arteriole-to-venule ratio, and the number of capillaries per 1mm2 of conjunctiva in former smokers were associated with cessation of smoking.
Today, researchers regard cessation of smoking as the most effective strategy for primary and secondary prevention of MI: In smokers, the risk for MI is three times higher than in former smokers. In former smokers, the risk for repeat infarction is similar to thatseen in non-smokers with no history of infarction [9]. In smokers with MI the pathophysiological mechanisms of arteriolar narrowing are probably the same as in apparently healthy young people who use tobacco [3,5,10]. However, in IHD patients MCB parameters are initially altered, which differentiates them from apparently healthy young people. The mean arteriolar diameter was 20.59 ± 0.63μm in young patients who had never smoked and 13.01 ± 0.41μm (the lowest value) in smokers. After patients quit smoking, this parameter significantly increased, up to 18.34 ± 0.65 [6].The same trends were observed in IHD patients. Analysis of the arteriole-to-venule ratio showed that the effects of smoking on MCB structure were similar in apparently healthy young people and IHD patients. So, the arterioleto- venule ratio was 0.61 ± 0.02 in healthy young non-smokers and 0.56 ± 0.02 in IHD patients who did not smoke tobacco. In healthy young smokers and IHD patients who smoked tobacco, this parameter was lower (0.39 ± 0.01 and 0.4 ± 0.02, respectively). In healthy young former smokers and IHD patients who had quit smoking, this parameter was significantly higher (0.5 ±0.02 and 0.56 ± 0.03, respectively) than in the group of smokers. In addition to natural and expected changes in MCB related to age and/or the underlying disease (MI in IHD patients), smoking causes further changes in the diameter and number of microvessels. After the cessation of smoking, these smoking-induced changes are reversed and MCB parameters return to their initial values, similar to those seen in non-smokers with similar other characteristics (age, medical condition, etc.).
Figures 1-3 illustrate these results by showing MCB in three patients with AMI: a patient who has not smoked a current smoker, and a former smoker, respectively.
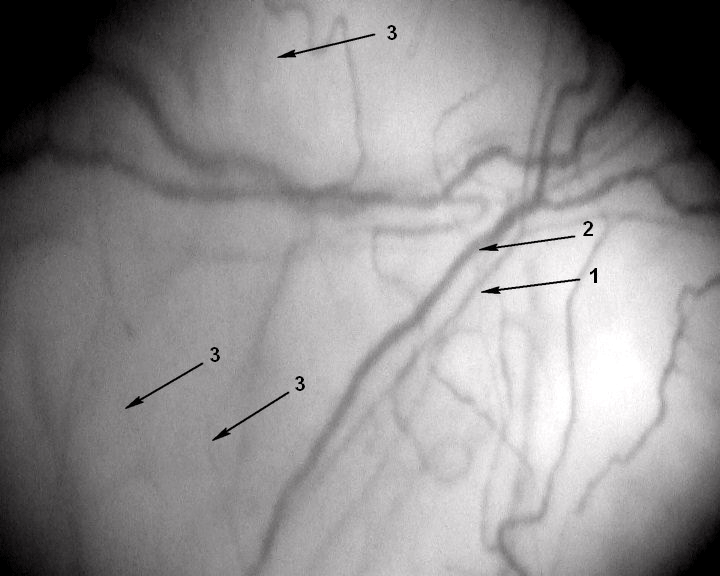
Figure 1: A segment of the microcirculatory bed. Magnification 96x.
Patient F, 62 years old, has never smoked with no past medical history. His
IHD was manifested as a Q-wave MI. The patient was examined on day 10
of in-hospital treatment.1 – arteriole (d 16.7μm), 2 – venule (d 29.4μm), 3 –
capillaries (d 9μm); number of capillaries per 1 mm2 of conjunctiva – 4.1units/
mm2.
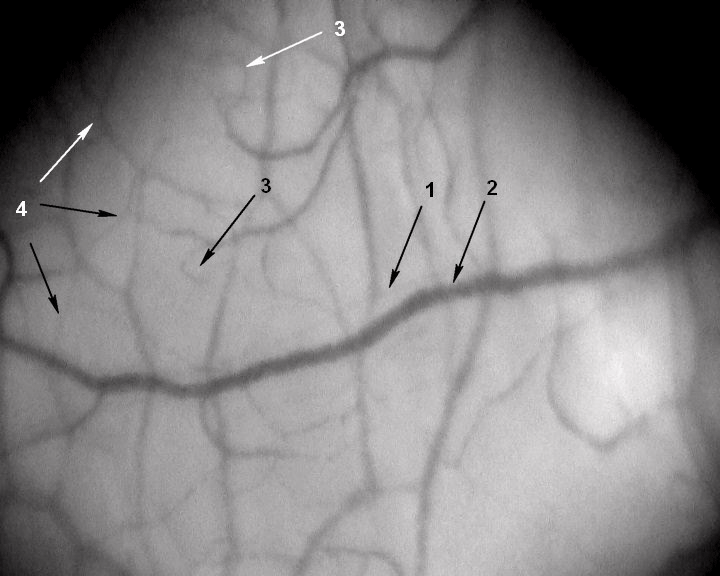
Figure 2: A segment of the microcirculatory bed. Magnification 96x. Patient Z,
45 years old, with no past medical history. His IHD was manifested as a
Q-wave MI. The patient was examined on day 10 of in-hospital treatment.
He has smoked for 39 years (15 cigarettes a day). 1 – arteriole (d≈ 5μm), 2 –
venule (d 32.1μm), 3 – capillaries (d 8.8μm); number of capillaries per 1mm2
of conjunctiva – 3.5unit/mm2; 4 – network of newly formed vessels.
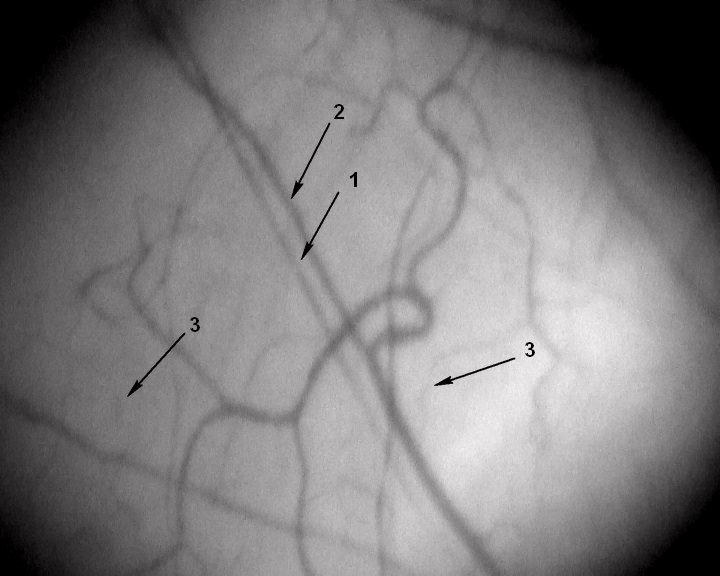
Figure 3: A segment of the microcirculatory bed. Magnification 96x.
Patient K, 57 years old, a former smoker with no past medical history. His IHD
was manifested as a Q-wave MI. The patient was examined on day 10 of inhospital
treatment. He smoked for 20 years (20 cigarettes a day), but stopped
10 years ago. 1 – arteriole (d 21.1μm), 2 – venule (d 26.2μm), 3 – capillaries
(d 9.1μm); number of capillaries per 1mm2 of conjunctiva – 4.4unit/mm2.
Conclusion
In former smokers in the acute stage of MI, the mean arteriolar diameter, the mean capillary diameter, the arteriole-to-venule ratio, and the number of capillaries per 1mm2 of conjunctiva were significantly higher than in current smokers, but these numbers did not differ from those found in the non-smoking group. Thus, smoking adversely affects the structure of MCB, which can recover after the cessation of smoking. The positive changes in the MCB of former smokers with IHD support the recommendation that patients with this condition stop smoking.
References
- Kireev KA, Fokin AA, Kramnik GE. Smoking prevalence in acute myocardial infarction patients with sudden death risk. Cardiovascular Therapy and Prevention. 2017; 16: C. 28-32.
- Korneeva NV. The simplified algorithm of the video biomicroscopy of a bulbar conjunctiva in the young quitted smoking subjects by means of the logistic regression method. Regional blood circulation and microcirculation. 2016; 15: 75-81.
- Korneeva NV, Sirotin BZ. Microcirculatory bed, microcirculation, and smokingassotiated endothelial dysfunction in young adults. Bulletin of experimental biology and medicine. 2016; 162: 800-804.
- Lang TA, Secic M. How to Report Statistics in Medicine: Annotated Guidelines for Authors, Editors, and Reviewers / Russian version, edited by V.P. Leonova. – M.: Prakticheskaya Meditsina (Practical Medicine). 2011; 480 p.
- Nikolaev K Yu, Lifshits GI, Pronin VS. The pathophysiological aspects of influence with chronic tobacco smoking on microcirculation system. Herald of the Novosibirsk State University. SERIES: BIOLOGY, CLINICAL MEDICINE. 2010; 8: 119-122.
- Sirotin BZ, Korneeva NV, Yavnaya IK. Microcirculatory bed in healthy young people after their discontinuation of tobacco smoking. Far East Medical Journal. 2015: 13-15.
- Sirotin BZ, Shevtsov BP, Davidovich IM, et al. Influence of smoking on the microcirculation of bulbar conjunctiva vessels in young patients with coronary artery disease. Therapeutical archive. 1982; 70-73.
- Sirotin BZ, Iavnaia IK, Zhmerenetskii KV. The microcirculatory bed in patients with coronary heart disease and its effect from cigarette smoking. Preventive Medicine. 2013; 16: 44-48.
- Serrano M, Madoz E, Ezpeleta I, San Julián B, Amézqueta C, Pérez Marco JA, et al. Smoking Cessation and Risk of Myocardial Reinfarction in Coronary Patients: a Nested Case-Control Study. Rev EspCardiol. 2003; 56: 445-451.
- Barua RS, Ambrose JA, Eales-Reynolds LJ, et al. Dydfunctional endothelial nitric biosynthesis in healthy smokers with impaired endothelium-dependent vasodilatation. Circulation. 2001: 1905-1910. doi: 10.1161/hc4101.097525.
