Review Article
Austin J Biomed Eng. 2014;1(2): 1007.
Emerging Optical Techniques for Detection of Oral, Cervical and Anal Cancer in Low-Resource Settings
Bing Yu1*, Daron G. Ferris2 and Yang Liu3 and Vivek Krishna Nagarajan3
1Department of Biomedical Engineering, University of Akron, USA
2Department of Obstetrics and Gynecology, Georgia Regents University, USA
3Department of Biomedical Engineering, University of Akron, USA
*Corresponding author: :Bing Yu, Department of Biomedical Engineering, University of Akron, Akron, OH 44325-0302, USA.
Received: March 20, 2014; Accepted: April 10, 2014; Published: April 14, 2014
Abstract
Cancers of the oral and anogenital regions are a growing global health problem that disproportionately impact women and men living in developing countries. The high death rate in developing countries is largely due to the fact that these countries do not have the appropriate medical infrastructure and resources to support the organized screening and diagnostic programs that are available in the developed world. Emerging optical diagnostics techniques, such as optical spectroscopy, reflectance imaging, and fluorescence imaging, are noninvasive techniques that are sensitive to multiple cancer biomarkers and have shown the potential as a cost–effective and fast tool for diagnosis of early precancerous changes in the cervix, oral cavity and anus. This paper provides a review of current strategies for prevention, screening and diagnostic tests of oral, cervical and anal cancers and development in optical diagnostic techniques that could potentially be used to improve current practice in resource–limited settings.
Keywords: Oral Cancer; Cervical Cancer; Anal Cancer; Low–Resource Settings; Optical Imaging; Optical spectroscopy
The global burden of HPV–related cervical, anal and oral cancers
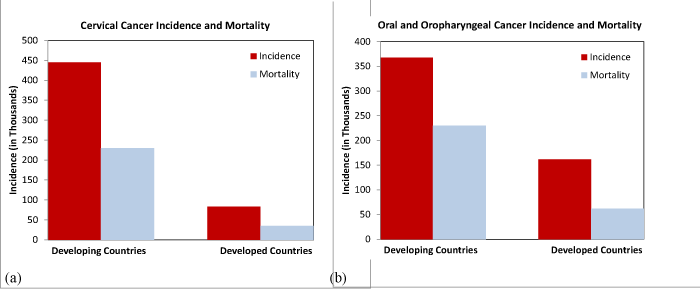
Each year there are more than 750,000 cases of anogenital and oropharyngeal cancers [1]. Of these human papillomavirus (HPV)–induced cancers, there are 528,000 cases of cervical cancer with 274,000 deaths per year (Figure 1a) [1]. The majority of these cases (approximately 85%) occur in resource–poor countries [1,2]. Worldwide, cervical cancer is the second most common cancer among women. The highest incidence rates of cervical cancer are found in Central and South America, Africa and Asia where rates exceed 50 cases per 100,000 women. Because of the lack of modern prevention practices in these areas, the global mortality rate of cervical cancer is estimated to increase by 25% during the next 10years [2]. While the Pap test has substantially reduced the rate of cervical cancer, a preventable disease, too many women continue to suffer even in resource–rich countries. More women’s life years (death at a younger age) are lost with cervical cancer compared withovarian or breast cancer. Although less than 15,000 women in the United States develop cervical cancer per year, greater than ten–fold more are afflicted by precancerous conditions causing significant morbidity from additional diagnostic tests, histological sampling and surgical interventions. This toll occurs despite compliant surveillancepractices in resource–rich nations. Yet, these secondary cancer prevention strategies are associated with tremendous expense ($8 billion⁄yr. in US), complicated logistics and significant morbidity.
Figure 1: Global cancer incidence and mortality rates for: (a) cervical cancer and (b) oral and oropharyngeal cancers. (Data extracted from the GLOBOCAN2012).
Each year, more than 650,000 new cases of head and neck cancer are diagnosed worldwide, of which over one–third are oral or oropharyngeal cancers [3,4]. As such, cancers of the head and neck are the eighth most common type of cancer. Of these oral cases, nearly two–thirds occur in resource–poor countries (Figure 1b) where diagnostic and therapeutic interventions are limited or nonexistent. Tobacco and alcohol use are the main risk factors for head and neck cancer. Consequently, men are at greater risk for this cancer. However, the vast majority of oropharyngeal cancers and many oral cancers are caused by infection with oncogenic HPV.Moreover, the rate of HPV–induced oropharyngeal cancers is rising at an alarming rate. Certain sexual practices have been implicated for this increasing trend. But these cancers are extremely challenging to detect. Approximately 90% of oropharyngeal cancers are buried in the crypts of the tonsils or located at the base of the tongue [5,6].Both of these sites are difficult to screen clinically due to location and the tendency to elicit an uncomfortable gag reflex in the patient. In contrast with HPV–induced disease located in other parts of the body, oropharyngeal precancer lesions are not visually appreciated. Consequently, virtually no screening testing is performed for oropharyngeal cancer, particularly in resource–poor regions. Becauseof no screening test, oropharyngeal cancers are usually detected only when a large mass is noted. At this late stage, therapeutic cure becomes exceedingly challenging. Despite various treatment options, the 5–year survival rate is approximately 25% [5,6].
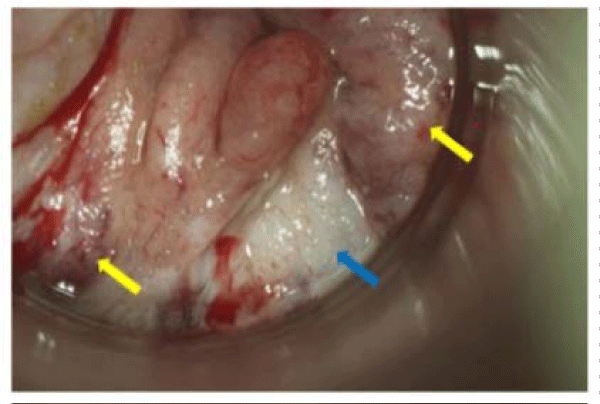
There were 30,400 cases of anal cancer diagnosed worldwide in 2002 [7]. In the United States, incidence rates of anal cancer and severe anal precancers have increased twofold since the 1970s [7]. There are approximately 6,000 new cases of anal cancer diagnosed annually in the US, the majority seen in women. Certain populations are at an increased risk for anal cancer including immune suppressed individuals (HIV+, organ transplant or other patients on immunosuppressive pharmacologic agents), patients at behavioral risk (men who have anal sex with men) and women with a history of severe HPV–induced genital precancers or cancer. Although difficult to estimate, there are likely 100 times more cases of anal pre–cancers than anal cancer. Much like cervical cancer, anal cancer is preceded by a pre–malignant condition providing an opportunity for early detection and treatment (Figure 2). However, screening capabilities or expertise in resource–poor countries are essentially nonexistent. Therefore, in contrast with other HPV–induced cancers, more than half of the cases of anal cancer are found in resource–rich nations. In the US, 80% of such cases are derived from squamous epithelium. Yet in Japan, 80% are of glandular cell origin which is more difficult to detect by current screening and diagnostic methods. Advanced cases of anal cancer have a high mortality rate depending on the surgical stage and nodal involvement [7]. Treatment regimens for anal cancer are complicated and often alter quality of life [8].
Figure 2: Anal cancer (indicated with yellow arrows) and precancer (indicated with blue arrow). The anal cancer is characterized by an irregular surface contour and the presence of atypical blood vessels. The epithelium of the precancer is thickened and opaque white (photo courtesy of Daron Ferris, MD).
Hence, HPV–related cancer remains a daunting problem for public health, health care providers, insurers, patients and society. The challenge is even greater for resource–poor countries.
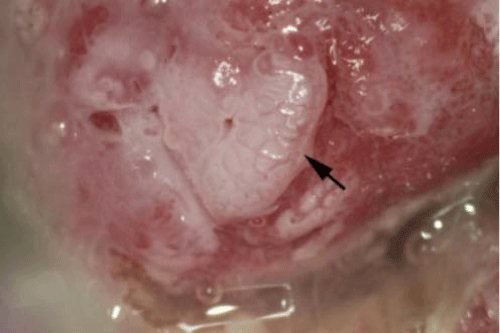
HPV is a highly contagious viral DNA organism and the most common sexually transmitted infection. Essentially ubiquitous, approximately 80% of individuals will become infected with HPV during their lifetime. The peak prevalence of HPV infection is between 15 to 25 years of age. While the majority of HPV infections regress spontaneously without treatment, particularly in immunecompetent individuals, with continued viral persistence some produce neoplasias. Facilitated by superficial trauma, HPV infects both squamous and columnar epithelium. More than 100 types of HPV exist with some being oncogenic and others considered nononcogenic. Based on regional tropism, approximately 40 HPV types infect only the epithelium of the anogenital and oropharyngeal tracts. Of these, approximately 16 are cancer causing types. HPV 16 is the most frequent HPV type detected in cervical (in addition to vaginal and vulvar), oral and anal cancers. HPV 16 and 18 cause approximately 70% of cervical cancer (Figure 3), 80–90% of anal cancer and nearly all cases of oropharyngeal cancers [4,9–14]. Approximately 50% of highgrade cervical lesions (Cervical Intraepithelial Neoplasia (CIN) 2/3) and 25% of CIN1 are caused by HPV 16 or 18. The remaining HPV types cause condyloma (warts), premalignant diseases or cancer. Infection may produce condyloma, precancers or cancer of the cervix, vagina, and vulva in women, penis in men, and oropharynx and anus in both sexes. Non–oncogenic HPV causes recurrent respiratory papillomatosis, a debilitating and sometimes lethal upper airway condition characterized by diffuse condyloma that may compromise respiratory function [15]. An oncogenic HPV infection of immature metaplastic epithelium in the cervical or anal transformation zone initiates pathogenesis. With HPV persistence, cervical or anal cancer precursor lesions or cancers may slowly arise [16]. While cervical cancer usually results from the serial progression of CIN1 to CIN2 to CIN3 and then cancer, some oncogenic HPV infections advance directly to CIN3. However, the overall gradual development of these neoplasias to cancer normally enables routine screening programs to detect disease at early, universally curable stages.
Figure 3: Cervical cancer (indicated by the yellow arrows) is seen on both the anterior and posterior cervix in a 24 year old woman. The epithelial surface is elevated, friable and yellowish in color. Atypical blood vessels are noted, the hallmark of invasive cervical cancer. This cancer was caused by HPV 16 (photo courtesy of Daron Ferris, MD).
Current strategies in prevention, screening and diagnostic tests
Prevention– HPV vaccines
Using recombinant DNA virus–like–particles (VLPs) that cannot convey genital infection, scientists have developed several human papillomavirus (HPV) L1 VLP vaccines. The quadrivalent HPV (qHPV) 6, 11, 16 and 18 L1 VLP vaccine (Gardasil®, Merck and Co, Inc, White Station, NJ) and a bivalent HPV 16 and 18 L1 VLP vaccine (Cervarix®, GSK, Rixensart, Belgium) prevent approximately 70% of all cervical cancers and the qHPV vaccine also prevents 90% of anogenital warts. These two prophylactic HPV vaccines greatly diminish the morbidity and mortality associated with HPV– induced neoplasias of the anogenital tract [17–23]. The qHPV vaccine is 100% effective in preventing anogenital neoplasia associated with the four HPV vaccine types when administered to people naive to those HPV types. Early adolescents (ages 11–12) who are not yet sexually active represent the ideal target age for vaccination. However, the qHPV may be given intramuscularly in 3 doses to both boys and girls ages 9–26. In addition to excellent efficacy, multiple clinical trials and National Adverse Event surveillance systems (CDC Vaccine Adverse Event Reporting System (VAERS) and CDC Vaccine Safety Data link (VSD)) have demonstrated good qHPV vaccine safety. Studies have determined HPV vaccination to be cost effective. The best cost saving strategy occurs with maximum vaccine uptake. Widespread gender neutral HPV vaccination also critically impacts HPV–related disease. Both HPV vaccines are prophylactic and do not convey therapeutic benefit. A novel nine–valent HPV vaccine is currently under review by the FDA. This vaccine is expected to prevent 90% of cervical cancer cases.
Great enthusiasm surrounded the advent of these two novel primary cancer prevention vaccines. These prophylactic vaccines will provide tremendous benefit by reducing the mortality and morbidity associated with HPV–induced cervical cancers. Protection from HPV–induced neoplasia is also afforded to the remainder of the lower genital tract, including the vagina, vulva, perianal and anal region.In addition, vaccination is expected to reduce the rate of head and neck cancer and recurrent respiratory papillomatos is that are caused by high risk and low risk HPV types, respectively. But despite extensive marketing of the HPV vaccines in the United States and other resource rich countries, vaccination rates remain embarrassingly low. Poor qHPV vaccine uptake in the United States (33% for all 3 doses in 2012) has even raised concern from the Centers for Disease Control and Prevention (CDC) [24]. Vaccine uptake is lowest in the southeast United States where the highest rates of cervical cancer mortality are also found. However, some countries using mandatory, school based vaccination programs (i.e., Australia) have already benefitted from the vaccine [25–28]. Yet, even there, the current HPV vaccines will reduce but not totally eliminate HPV–induced cancers. Unfortunately, distribution of HPV vaccines to resource–poor regions has suffered from high cost and logistical issues. Hence, because of slow vaccine uptake worldwide, a large number of unvaccinated populations are exposed to the risk of other HPV types, traditional screening programs are still essential.
Screening
Papanicolaou test: The Papanicolaou (Pap) smear is the most cost effective cancer screening test ever introduced. Where effectively implemented, rates of cervical cancer have decreased by more than 70%. While screening guidelines vary worldwide, generally women between 21 and 65 years of age are screened. Routine anal Pap screening is also recommended for high–risk individuals [29–31]. The test involves careful sampling of the uterine cervical or anal transformation zones (squamous ectocervical, columnar endocervical, and interposed metaplastic cells) using one or more cytologic collection devices. Two types of Pap tests exist, the conventional Pap test and liquidbased Pap tests. While the collection methods are identical, with the former test, cells are transferred directly to a glass slide then quickly fixed at bedside in the exam room. Using liquid–based testing, after sampling, the cells are instead transferred to a liquid preservative and the slide is later processed in the laboratory. Both types are Pap stained and then examined microscopically by trained health care providers. Automated, computer–based technology is also used to examine Pap tests for quality control (re–read of negative test results) or primary screening purposes. Cytologic diagnoses are rendered using the Bethesda System terminology as NILM, ASCUS, ASC–H, AGC, LSIL, HSIL, AIS, and cancer. The conventional cervical Pap testis approximately 50% sensitive at detecting cervical neoplasia and the liquid–based test is approximately 80% sensitive. Using residual cells, the latter collection method also permits co–testing for oncogenic HPV either as a reflex triage test for women with Atypical Squamous Cells of Undetermined Significance (ASCUS) results or in primary screening (co–testing) with the Pap test for women greater than 30 years of age. The sensitivity of the anal Pap test is much lower and the rate of unsatisfactory tests much greater due to limitations of the Dacron swab used for anal Pap testing. Clinician sampling errors are the main cause for failure to detect disease.
While the Pap test has decreased the rate of cervical cancer where effectively implemented, it has not been successfully introduced in resource–poor countries. There, available programs are opportunistic and not well organized. Due to the relative lack of sensitivity of the conventional Pap test, annual screening is necessary to detect precancers before developing into cancers. Cancer can be universally prevented when premalignant changes are detected early and treated adequately. However, compliant and accurate surveillance, along with proper diagnosis and treatment are required for this satisfactory outcome.
HPV testing
Bimolecular testing for the presence of oncogenic HPV has been incorporated into the cervical cancer screening and triage programs in some resource rich nations [32–37].The reason for the change is due to the relative insensitivity of the Pap test, the discovery of oncogenic HPV as the causal agent for cervical cancer and supportive results from clinical trials. Multiple HPV DNA and mRNA tests are commercially available to detect multiple types of oncogenic HPV or selective HPV types (i.e., HPV 16 and HPV 18). According to American Society for Colposcopy and Cervical Pathology (ASCCP) management guidelines, there are several uses for HPV testing [38]. HPV testing can be used either as a reflex triage test for women with a typical Squamous Cells of Undetermined Significance (ASCUS) results, as a triage test following therapy of HPV–related neoplasia, in primary screening (co–testing) with the Pap test for women greater than 30 years of age or perhaps as a single HPV multi–test for screening purposes. Based on its very high negative predictive value (99%) for excluding significant cervical neoplasias, there is growing confidence in this test. When used as a primary screening test, greater than 90% of all cases of CIN2/3 or cancer are detected [39]. Primary screening with HPV tests is also approximately 25% relatively more sensitive than cytology at detecting neoplasias. Unfortunately, the positive predictive value of a single HPV test result for predicting the presence of severe cervical neoplasia correctly is no greater than 20%. In many cases, a positive test may simply indicate acute, subclinical HPV infection (no detectable dysplasia), transmissible HPV from a sexual partner or only a transient condition. Because the prevalence of HPV is quite high in women 30 years of age or younger, its use is limited primarily to older women.
Most HPV tests require sophisticated laboratory testing and trained lab technicians. Furthermore, the tests are relatively expensive and may not include all of the identified oncogenic HPV types. However, because HPV DNA testing is more sensitive than Pap testing, there may be a future role for affordable, simple, point–of–care HPV tests in resource–poor locations [40].
Visual inspection with acetic acid (VIA)
Visual Inspection following Acetic acid application (VIA) consists of a simple, contrast solution–enhanced visual inspection of the cervix without use of special equipment [41–43].Cervical epithelial cells with a high nuclear to cytoplasmic ratio (normal immature metaplasia and all neoplasias) briefly appear white after exposure to 5% acetic acid (vinegar). Following a one minute application of undiluted vinegar, any resulting transient white epithelium noted on the ectocervix by naked–eye examination is considered abnormal. Absence of a white color or a normal pink/red color is considered a negative test result. Individuals with limited medical experience can be trained to perform VIA exams. Little equipment or supplies are needed, only vinegar, cotton applicators, a light source and vaginal speculum. Visual Inspection with Lugol’s Iodine (VILI) is similar, except 50% diluted Lugol’s iodine is applied to the ectocervix of women not allergic to iodine [44]. Normal native squamous epithelium and mature metaplasia contain glycogen in the superficial and intermediate cell layers. Iodine has an affinity for glycogen; hence normal epithelium appears mahogany brown following the application. Normal immature metaplasia and all neoplasias do not contain glycogen, and therefore appear yellow following iodine application. Hence, a brown color is considered a negative result and yellow a positive VILI result. Improved sensitivity can be obtained if both VIA and VILI are used concomitantly [44]. Because no special equipment is necessary, VIA and VILI are considered inexpensive screening tests for cervical cancer. They have proven somewhat effective at reducing cervical cancer rates in resource–poor countries where no other screening options are available. The sensitivity of VIA is similar to the Pap test, but unlike the Pap test, it does not require specimen collection or processing. Yet, there is substantial variability in the sensitivity of VIA primarily due to experience, training and inherent weaknesses of the test. According to some, the most efficient and effective strategy to prevent cervical cancer in low–resource settings is to screen using HPV testing or VIA, followed by treatment of the pre–cancerous lesions using cryotherapy (freezing) [45,46]. This should optimally be carried out in a single visit by physicians, nurses or midwives. Until low cost HPV testing is realized, VIA or VIA with low power magnification (VIAM) combined with cryotherapy may be the most viable option for reducing the mortality associated with cervical cancer in the developing world.
However, in a large study in India, HPV testing was associated with a significant reduction in the number of women with advanced cervical cancers and cervical mortality when compared with Pap testing and VIA.[45]Others have also demonstrated the sensitivity of VIA to be much less than the sensitivity of HPV DNA testing [41].This inherent weakness of VIA and VILI is due to the same nonspecific color reactions that occur in both normal and neoplastic epithelium. The poor test specificity of VIA leads to many normal women, particularly young women with an active transformation zone (appears white with VIA testing), receiving unnecessary treatment. VIA is also unable to examine the endocervical canal where many neoplasias arise, particularly in postmenopausal women or women using progesterone only contraception. As a consequence, VIA suffers from the inability to determine exam adequacy (identification of the entire squamo columnar junction and transformation zone), discriminate epithelial and vascular changes, appraise positive findings using intense illumination and magnification, and recognize falsely positive diagnoses secondary to the presence of immature metaplasia, repair or inflammation that also assumes a transient aceto white, or in the case of VILI, a yellow color.
Diagnosis–colposcopy and biopsy
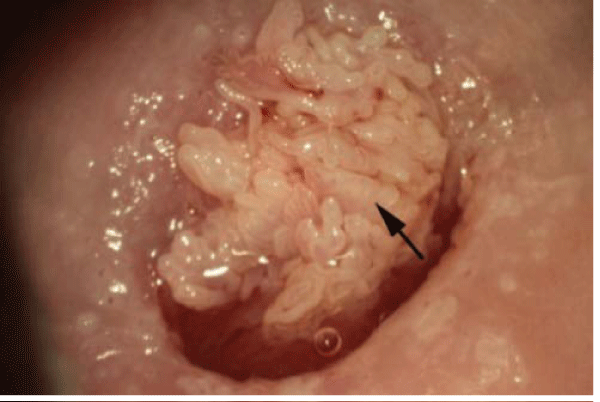
Cervical cancer can be universally prevented when certain premalignant lesions of the cervix are detected early and treated adequately. However, compliant and accurate surveillance, along with proper diagnosis and treatment are required for this outcome. Colposcopy is the diagnostic procedure used following screening tests to evaluate women with potential lower genital tract neoplasias (Figure 4 and 5). The colposcope, astereoscopic binocular microscope with a powerful light source, is used to detect cervical neoplasias and to identify abnormal tissue for biopsy [16].The goal of colposcopy is to identify cervical cancer precursors to enable eradication of premalignant lesions in order to preclude their progression to cancer. Cervical neoplasia exhibits certain morphologic features that can be identified during a colposcopic examination [16].Findings from the colposcopy exam and targeted biopsies determine subsequent patient management.
Figure 4: Cervical condyloma (indicated by the arrow) detected during colposcopy. The long villous-like projections are a common morphologic appearance of HPV-induced lesions(photo courtesy of Daron Ferris, MD).
Figure 5: Cancer precursor (indicated by the arrow)seen by colposcopy. The opaque white epithelium and coarsely dilated mosaic blood vessels are diagnostic of CIN 3(photo courtesy of Daron Ferris, MD).
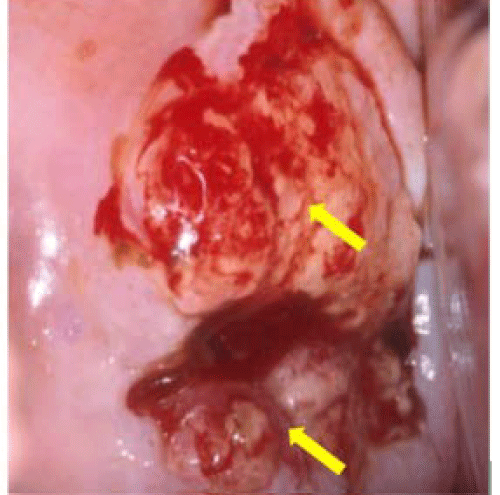
Multiple cervical lesions of varied severity are frequently seen at the same time and in the same patient (Figure 6). Normal anatomical variants also mimic true disease. Discriminating levels of colposcopic abnormality, and even normal from abnormal colposcopic findings, requires ample clinical experience. Determining where to selectively obtain small representative biopsies that reflect the worst disease is a skill only acquired with rigorous training. However, colposcopy is somewhat subjective and the quality of practice varies considerably. Recent evidence suggests an urgent need to improve the accuracy of colposcopy [47–55]. Without other viable solutions, it has been recommended that multiple biopsies be obtained during colposcopy to improve disease detection [56]. Yet, the collection of multiple randomly obtained biopsies is also fraught with serious problems. Proper colposcopy and cervical biopsy techniques are imperative during the evaluation of women with cervical cytologic abnormalities. Accurate cervical biopsy is the principal factor that ultimately determines proper diagnosis and management. However, cervical neoplasias identified by random biopsy may not actually represent true cancer precursor lesions that require surgical intervention as small, occult lesions may resolve spontaneously. Further, a random biopsy approach risks histological overcall of normal epithelia (immature metaplasia) that mimic neoplasia. Subsequent treatment would be unnecessary. Hence, there may be a role for sophisticated novel technology to assist the identification and assessment of cervical lesions.
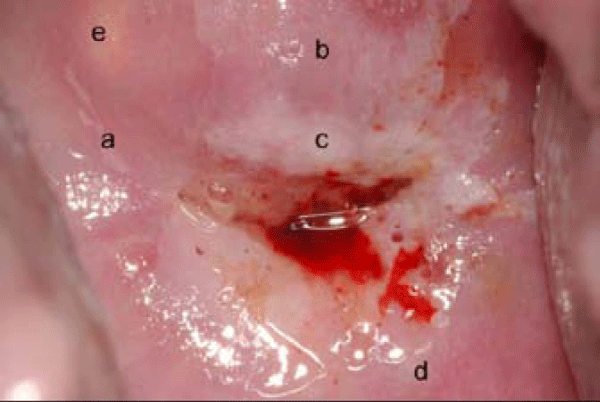
Figure 6: Multiple potential biopsy sites risks improper patient management. This case depicts the potential ambiguity in selecting the most severe area of neoplasia. Both a (small satellite lesion) and b (large 2 quadrant lesion) represent CIN1. Because b is readily apparent, most colposcopists would biopsy this site. An area of subclinical HPV infection is seen at d. The yellow area e with dilated vessels mimics cancer, but merely represents a benign Nabothi an follicle. The smaller lesion c is a true cancer precursor (CIN3). If biopsies were incorrectly obtained from a, b, d or e, the patient would receive no treatment. However, if c is biopsied, surgery is required to prevent progression to invasive cancer (photo courtesy of Daron Ferris, MD).
Challenges of implementing current cancer prevention strategies in developing countries
Because cervical cancer is preceded by premalignant cervical intraepithelial neoplasia (CIN), if detected early and treated adequately, cervical cancer can be universally prevented. However, adequate resources for regular screening, appropriate diagnoses, effective treatment and proper follow–up are required. Unfortunately, for many women living in developing, resource–poor countries, modern screening methods designed to detect precancerous cervical disease are either not available or not affordable. Consequently, cervical cancer is the number one cause of death from cancer for women in these countries.
As previously discussed, the Pap test has been the gold standard cervical screening test for many years. Where implemented on a serial interval basis, cervical cytologic screening using conventional Pap tests has significantly reduced the incidence of cervical cancer [57,58]. Yet, the highly complex Pap test system suffers from sampling, laboratory and equipment errors, [59, 60] patient noncompliance, [61] inappropriate interval screening, [62,63] inadequate follow–up of abnormal Pap test results, [64,65] poor sensitivity, [66,67] and the need for multiple (annual) testing and multiple patient visits. Moreover, because of certain subjectivity, Pap test inter–observer reproducibility is only fair (k=0.46) [68]. Liquid–based cervical cytology is more sensitive in detecting cancer precursors than a conventional Pap test. Yet, because it is considerably more expensive, requires complex laboratory processing and equipment, and may not actually reduce the incidence of cervical cancer in comparison with conventional cytology, its utility in disadvantaged populations is suspect. Hence, there has been a limited role for cervical cancer screening using a Pap test in resource–poor regions. Moreover, when adopted in many of these areas, the effectiveness has been disappointingly poor.
As a consequence of the limitations of both types of Pap tests, other cervical cancer screening techniques have evolved. HPV DNA and mRNA tests are more sensitive than the Pap test but less specific. Unfortunately, these tests are cost prohibitive for resource–poor nations. Moreover, testing is labor intensive, at risk to contamination (PCR processing) and technically too advanced for rural settings.
In regions with poor or nonexistent cervical cancer screening programs, the HPV vaccine offers the best option to reduce the morbidity and mortality associated with anogenital and oropharyngeal cancers. However, widespread implementation is required, in addition to a reliable cold–chain transport system. Presently, most resource–poor nations are critically challenged by the cost of vaccination and lack of appropriate infrastructure. Rapid global implementation is doubtful as evidenced even in resource rich countries. Given a delayed adoption of HPV vaccination worldwide, other novel means of cancer prevention are urgently needed. But certain impediments, somewhat unique to resource–poor countries, also challenge current HPV–induced cancer prevention efforts.
In resource–poor settings, barriers to cervical cancer screening include lack of access to health care, shortage of quality medical centers and laboratories, few trained and experienced personnel, distance to health care clinics, irregular or inconvenient hours of operation, long waiting times, and lack of affordable options for follow–up care (Figure 7). Individual and cultural barriers include lack of a tradition of preventive health seeking behaviors, less exposure to medical procedures, poor knowledge of cervical cancer, language barriers, negative views of health care providers, lack of spousal support for health care, anxiety and fear over potential serious diagnoses and cancer, shortage of female healthcare providers, and modesty over exposing the genital region [69]. Other factors include religious beliefs, perceived lack of risk, embarrassment, and other competing priorities (lack of time). Failure to obtain cervical cancer screening is the major reason for developing cervical cancer (other than failure to clear oncogenic HPV infection once infected). In fact in the United States, 50% of all cases of cervical cancer are detected in women who never have received adequate screening. The reasons for high cervical cancer mortality rates are listed below:
Figure 7: Women awaitingmodern liquid-based Pap testing by CerviCusco in the Andes Mountains of Peru (photo courtesy of CerviCusco, Peru).
- Nearly nonexistent and otherwise ineffective Pap test screening programs
- Inability to afford a Pap test
- Low priority for Ministry of Health — Competing health problems
- No national laboratory quality control or standards
- Lack of properly trained personnel
- Few modern materials, supplies and equipment
- Absent infrastructure
- Complex cancer prevention programs difficult to coordinate
- Unreliable power supply
- Delay in Pap test results leading to noncompliance (loss to follow up)
- Paucity of experienced health care providers
- Minimal access to care for screening, diagnosis and treatment
- Inadequate patient education
- Most women diagnosed with advanced cervical cancer when few treatment options are available and survival rates are poor
Cervical cancer prevention is thus extremely challenged in developing nations. However, anal cancer screening is virtually nonexistent in resource–poor countries. Oral cancer screening involves a rudimentary visual inspection and clinical palpation. Yet, even in developed countries, this practice does not prevent oral cancer, but only helps in early stage detection. Hence, the high mortality rate of HPV–induced cancers in developing countries is largely due to the fact that these countries do not have the appropriate medical infrastructure and resources to support the organized screening and diagnostic programs that are available elsewhere. Despite advances in the prevention of cervical, anal and oral cancer with the introduction of the HPV vaccine and attempts to deliver comprehensive cervical, anal and oral cancer screening programs, women from developing countries continue to experience health disparities for cervical cancer morbidity and mortality. These health disparities between women from developed and developing countries signal the need for innovative approaches to reduce these disparities and improve the quality of care and health. An accurate, well tolerated, widely accessible, relatively inexpensive, simple and rapid on–site diagnostic instrument to screen and triage women for anogenital and oral neoplasias could reduce the incidence of cervical, anal and oral cancer, particularly for inhabitants in resource–poor nations. Hence, because of the many challenges to HPV–related cancer prevention described, optical diagnostic techniques are necessary and may potentially solve many of the current existing dilemmas encountered in the effort to reduce these preventable cancers.
Optical diagnostics
Numerous optical diagnostic techniques have been developed in the last two decades for the detection of epithelial cancers, such as those in the oropharynx and anogenital regions. These techniques include optical coherence tomography (OCT), reflectance imaging, diffuse reflectance spectroscopy (DRS), fluorescence imaging, and smart phone based diagnostic devices. Here we briefly review OCT and reflectance imaging, but focus our discussions on DRS, fluorescence imaging, and smart phone based diagnostic devices forthe potential in resource–poor settings.
OCT
OCT is a new optical technology for performing high–resolution, noninvasive cross–sectional imaging in biological systems [70]. It uses low–coherence interferometry to produce a 2D image of tissue microstructures in a way that is analogous to ultrasonic imaging. OCT was originally developed for retina imaging, but has recently been extended for oral, cervical and rectal cancers. Lee et al. [71] acquired OCT images of normal and precancerous oral mucosa and demonstrated to the use of the thickness of epithelium to achieve a sensitivity and specificity 82 and 90%, respectively. A literature review by Ovari et al., [72] indicated that OCT is a reliable method for identifying and targeted biopsy of inflammated, pre–malignant or cancer tissue in human laryngeal and pharyngeal mucosa. Liu et al., [73] investigated the use of OCT as an adjunct to colposcopy in the detection of CIN 2+ cervical neoplasia in a real–time clinical evaluation and increased the specificity from 83% to 93%.Gallwas et al., [74] evaluated the accuracy and reproducibility of OCT in the characterization of cervical intraepithelial neoplasia (CIN) and to distinguish between different CIN grades. They concluded that OCT is highly sensitive in identifying pre–invasive and invasive cancer of the uterine cervix. Zhang et al., [75] quantified the scattering coefficient of OCT images of rectal tissue by curve fitting the OCT signals to a confocal single model and demonstrated it as a promising diagnostic criterion of early rectal cancer. While OCT can provide high resolution 2D or 3D images of tissue structure and has significantly improved retina imaging, its application for oral, cervical and anal cancer detection in resource–poor settings is limited due to its high cost ($60,000 to $80,000) [76] and the need of experienced physician to interpret the images.
Reflectance imaging
Reflectance imaging, such as confocal microscopy and spectral imaging has found applications in oral, cervical and anal cancer screening and detection. Clark et al., [77] characterized features of normal and neoplastic oral mucosa in 15 biopsy pairs from 17 patients using reflectance confocal microscopy. Carlson et al., [78] used confocal reflectance microscopy on ex vivo cervical biopsies and in vivo cervical tissue to image the morphologic and biochemical changes associated with precancer. Wang et al., [79] demonstrated a MEMS micromirror enabled handheld confocal imaging probe for portable oral cancer detection with a large field of view. Roblyer et al., [80] designed a multispectral digital microscope with fluorescence, narrow–band reflectance, and polarized reflectance modes as a tool to improve detection of oral neoplasia. Subhash et al., [81] developed a diffuse reflectance imaging system which captures monochrome images of a patient’s mouth at 545 and 575 nm that were used to identify the margins of an oral lesion during invasive surgical interventions. Reflectance imaging system can potentially be made portable and cost–effective so that it could be used in developing countries [79]. However, remote image interpretation may be required to achieve an accurate diagnosis due to the lack of experienced imaging specialist in low–resource regions.
DRS
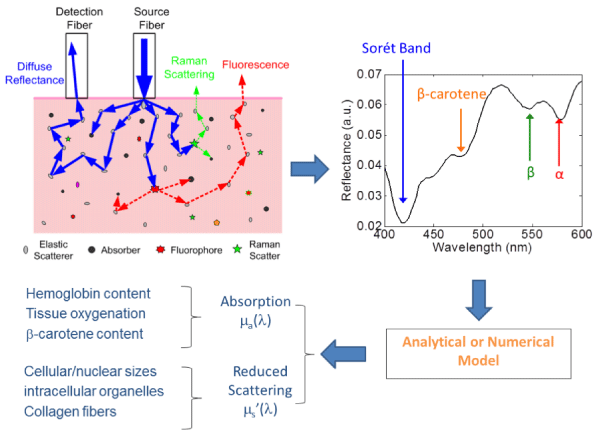
DRS, also known as elastic scattering spectroscopy (ESS), is a non–destructive technique designed to measure the absorption and scattering properties of turbid media, such as biological tissues [82– 92]. In DRS, a beam of broadband light emitted from a light source, such as thermal lamp, white LED or laser diodes, is launched into a tissue, often through multimode optical fibers, as illustrated in Figure 8 (upper left). The photons propagating in the tissue may experience various events, including elastic scattering, Raman scattering, absorption and fluorescence. Some of these photons escape from the tissue surface, after multiple elastic scattering, as reflectance. A detector, often a multimode optical fiber or fiber bundle, collects a portion of the reflectance and relay it to anoptical spectrometer, where the photons are converted to a wavelength–dependent intensity distribution of electrons, termed diffuse reflectance spectrum (upper right). The reflectance spectrum is analyzed using a model of photon propagation in tissue (e.g., diffusion equation, [82,93,94] Monte Carlo simulation, [95–98] or empirical model.[99]) to extract the absorption and reduced scattering coefficients (µa(λ) and µs’(λ)), as shown in Figure 8 (lower right). From the absorption spectrum, theconcentrations of the absorbers (such as oxy–hemoglobin, deoxyhemoglobin, beta–carotene and melanin, etc.) can be computed usingthe Beer–Lambert law (also known as Beer’s law).The scattering reflects the tissue morphological information, such as nuclear size and density. Both tissue compositions and morphological information have been identified as useful biomarkers for cancer diagnostics.
Figure 8: Illustration of the principle of diffuse reflectance spectroscopy with a fiber optic probe and the spectrum analysis procedures [100]. (Permission to reuse to be obtained from OSA).
DRS in the visible wavelength range (VIS–DRS) is sensitive to the absorption and scattering properties of epithelial tissue and has shown promise for early diagnosis of cancers in the cervix and oral cavity [101– 116]. The absorption and scattering properties of epithelial tissues reflect their underlying physiological and morphological properties [117]. In the visible band, dominant absorbers in oral and cervical tissue are oxygenated (HbO2) and deoxygenated hemoglobin (Hb), arising from blood vessels in the stroma. Light scattering is primarily caused by cell nuclei and organelles in the epithelium and stroma, as well as collagen fibers and cross–links in stroma. Neoplastic tissue exhibit significant changes in their physiological and morphological characteristics that can be quantified optically: Stromal layer absorption is expected to increase with angiogenesis, whereas stromal scattering is expected to go down with neoplastic progression as extracellular collagen networks degrade [96,103,117–120]. Epithelial scattering has been shown to increase due to increased nuclear size, increased DNA content, and hyperchromasia [96,117,118,121]. VISDRS have a penetration depth that can be tuned to be comparable to the thickness of the epithelial layer or deeper to probe both the epithelial and stromal layers [109,117,122]. Therefore, VIS–DRS have a great potential to be used as a cost–effective, fast and sensitive tool for diagnosis of early precancerous changes in the oropharynx and anogenital regions.
Current DRS systems typically consist of a broadband source, a spectrometer for multi–spectral detection and a fiber–optic probe for relaying light to and from the instrument [123]. The fiber–optic probe allows for contact measurement which provides access to many internal organ sites and a well–defined penetration depth. Contact measurement can also avoid specular reflection from the tissue surface which does do not carry any useful tissue information. However, the contact DRS systems have not been specifically designed to be robust and reliable, especially in resource–poor settings. Potential sources of systematic or random errors can arise from the uncontrolled probe–tissue interface and the lack of robust, real–time calibration technique. The lack of a robust, real–time calibration technique makes the calibration process time–consuming and potentially inaccurate, particularly when attempting to quantify absolute absorption and scattering coefficients. The uncontrolled probe–to–tissue coupling and pressure can make it difficult to obtain a reproducible and intact tissue reflectance spectrum. It is generally believed that the changes may be attributed to the compression of the blood vessels which causes reduced blood flow and alterations in the metabolism of the tissue as well as a change in the density of the scatterers [124–132]. It is therefore critical to measure and control the probe contact pressure in order to obtain reproducible and intact tissue physiological parameters.
More importantly, most VIS–DRS systems use thermal light sources, grating spectrographs, and cooled CCD cameras. Thermal light sources have large footprint, short life–time, low power efficiency, and low coupling efficiency to optical fibers. Spectrometers using grating spectrographs and cooled CCD cameras have extremely high wavelength resolution and sensitivity, but are very bulky and expensive and consume a large amount of electrical power. In addition, a stable power supply is very often required to operate a thermal lamp and a CCD camera. The system complexity also makes it necessary for the operator to have extensive knowledge in optical spectroscopy and professional training on the instrument and probes. Taken together, it is very difficult for VIS–DRS systems in their current forms to be directly used for cancer screening in resourcelimited settings.
Yu et al. recently reported the development of a smart fiber–optic probe based on a portable DRS instrument, shown in Figure 9, for oral and cervical cancer detection in developing world [100,133]. The smart fiber–optic sensor integrated a VIS–DRS channel, a self–calibration (SC) channel, and a diaphragm–based fiber–optic interferometric (DFPI) pressure sensor into a single fiber–optic probe to eliminate operator bias and to reduce size and power consumption as well as the need of operator training. The VIS–DRS channel measures the quantitative information about tissue physiological and morphological properties. The SC channel records a calibration spectrum in concurrent with the collection of a tissue reflectance spectrum. By dividing the tissue reflectance spectrum by the SC spectrum, the sources of errors due to light source drift and fiber bending loss can be removed in realtime. The pressure sensor provides real–time feedback on the probe pressure so that the operator can manually adjust the force applied on it. The device also used state–of–the–art photonics components, such as LEDs and miniature spectrometers, to reduce the size and power consumption, and automated Lab VIEW software to reduce the need of operator training. Therefore, it has a great potential as an accurate, fast, cost–effective and robust tool for detection of oral, cervical and anal cancers in resource–poor settings. The device has been tested for characterizing pressure response of oral mucosal tissue in vivo in healthy volunteers. Clinical validation of the device in a low–resource setting is necessary for identifying contrasts in optical biomarkers that vary with different grades of cervical intraepithelial neoplasia from normal cervical tissues.
Figure 9: (a)Photograph of the smart fiber-optic sensor system [100] (Permission to reuse to be obtained from OSA); and (b) a demonstration how the smart sensor is used with a speculum for cervical cancer detection.
Fluorescence imaging
Fluorescence imaging is a powerful spectro chemical method that has been widely used for imaging of endogenous fluorophores, such as Collagen, FAD and NADH, or contrast agents (or photosensitizers) in biological tissue. In fluorescence imaging molecules of the analyte are excited by light at a certain wavelength and emit light of a longer wavelength. Fluorescence imaging features very low detection limits and high molecular sensitivity and specificity. A number of studies have investigated the use of fluorescence imaging for screening and detection of cancers in the oropharynx and anogenital regions [118,134–138]. Shin et al., [139] reviewed recent advances in fluorescence imaging techniques to detect oral cancer and its precursors, while The kkek et al., conducted a detail review on optical imaging, including fluorescence imaging, for cervical cancer detection as a global problem [140]. Here we focus on two emerging low–cost imaging solutions, a high resolution micro endoscope (HRME) and a fluorescence goggle.
HRME
In 2011 Pierce et al. [141] reported the development of a low–cost HRME that can collects fluorescence images through a fiber–optic imaging bundle. The HRME employs a blue LED as its excitation source, a fiber–optic imaging bundle with 30,000 pixels for tissue illumination and fluorescence collection, and a CCD camera for image detection. It was claimed that the system can be built in a day using commercial components for under US$5,000. Pierce et al. [142] have conducted a pilot clinical study in China in 2012 to evaluate the HRME system for identifying women with cervical precancers. In the 174 women enrolled in the study, 69 sites appearing abnormal on colposcopy among which 12 showed CIN2+ on pathology. By quantification of the nuclear–to–cytoplasm ratio from the collected HRME images all the 12 pathology abnormal sites and 38 of the remaining 57 pathology normal sites were classified. The HRME also accurately classified all positive sites for high–risk HPV DNA. For the HPV positive sites which were found either normal by colposcopy or showed < CIN2 on pathology, only 6⁄32 (18.8%) were classified as abnormal by HRME. The study suggested that evaluation of suspicious lesions by HRME may increase the efficiency of current see–and–treat programs for cervical cancer.
Wearable Fluorescence Imaging System
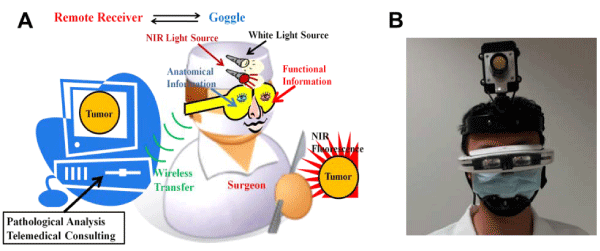
Liu et al. [143–146] developed a compact wearable near–infrared imaging system (fluorescence goggle) as an intraoperative tool for tumor detection and surgical guidance. Surgeons conventionally rely on pre–operative imaging technologies to guide surgical procedures. The system, as shown in Figure 10, is portable, small and userfriendly. Unlike conventional systems, the near–infrared fluorescence information is imaged in real time and displayed in the wearable eyepieces. Therefore, the point of view of the surgeon and that of the maging system are aligned. To achieve fast imaging without motion artifacts, CMOS sensor technology has been applied in fluorescence goggle systems.
Figure 10: Fluorescence goggle systems: (A) schematic illustration of the concept; [143] and (B) picture of a prototype fluorescence goggle system [144]. (Permission to reuse to be obtained from Elsevier and SPIE).
In conjunction with targeted molecular probes, it has been shown that fluorescence goggles accurately detect many forms of tumors including breast tumors and liver tumors, in small animal models [143–145]. Sub–millimeter nodules that are not obvious to unaided eye can be detected by fluorescence goggle. Recently, the prototype fluorescence goggle has been translated from the bench top into clinical surgical studies for hepatocellular carcinoma (HCC) [146]. The extension of tumors can be well visualized under the aid by florescence goggles [146]. In addition to guiding surgical resections, fluorescence goggles can also guide biopsy and staging procedures such as the sentinel lymph node biopsy. For instance, it can guide sentinel lymph node biopsy, a standard cancer staging procedure for many cancer forms [143,144,146]. The fluorescence goggles are compatible with many forms of contrast agents, including clinically approved indocyanine green and emerging agents such as quantum dots [143–146].
Studies have been successfully conducted to prove the feasibility of using the fluorescence goggle systems at the point of care [143]. For this purpose, the goggle has been integrated with wireless communication technologies. Using the wireless–capable fluorescence goggle, an experienced physician can guide the assessment of cancer from remote sites. In a preclinical study, the functional status of the tumor are imaged by a veterinarian performing the fluorescenceguided surgical procedures and simultaneously transferred wirelessly to the computer of a remote expert [143]. The video was displayed in real time, enabling remote assistance. Based on the data transmitted, the remote expert interacted verbally in real–time with the veterinary surgeon in the lab through a mobile phone to guide the surgery. Tumors were successfully located and removed under the wireless collaboration and the functional information of tumor status was stored at a remote computer. While the reported study simplifies clinical conditions, it demonstrates the potential of fluorescence goggles for telemedicine and point–of–care applications.
The fluorescence goggle platform has been applied to guiding breast and liver cancer surgeries, which showed the feasibility of using the system to guide oncologic interventions. In the future, the fluorescence goggle technology can be potentially applied to management of cervical, anal and oral cancer, in both developed countries and developing countries. The fluorescence goggles are cost–effective solutions for low resourced settings. It has been reported that prototype goggles can be constructed at a cost as low as $1,200. The affordable nature of fluorescence goggles renders them promising candidates for the screening as well as an intraoperative tool in the treatment of oral, cervical and anal cancers in rural areas and developing countries. They also hold great potential for expanding the current reach of image–guided interventions to currently underserved areas.
Smartphone based diagnosis
Smartphones represent one of the most exciting consumer electronics devices that have significantly changed the way people communicate during the past decade. Smartphones today, offer enormous computation power on a very compact platform. Moreover, they are integrated with some key features such as wireless communication, camera, global positioning systems (GPS), gyros, and accelerometers etc., which improve its functionality in an integrated manner. As a direct result of the versatility of smartphones, they are also playing a growing role in optical imaging for medical and biological applications, such as for spectroscopy, [147–150] microscopy, [149,151] counting molecules, [152] fluorescence imaging of single nanoparticles and viruses, [153] and cell–monitoring platform through lens less imaging [154].
The cost of wireless technology has also decreased over the years, thereby making smartphones a more affordable device. The number of smartphones has increased tremendously throughout the world in the last 5 years. Based on an UN report, there are currently 6.8 billion smartphones subscribers all over the world. More importantly, the report claims that the rate of cell phone diffusion into developing countries is at 89% as compared to 96% globally. This suggests smartphone–based diagnosis could potentially reduce healthcare costs, provide access to advanced laboratories through wireless communication to remote parts of the world, and be a fielddeployable means for diagnosis, etc. Smartphones in healthcare could revolutionize the field of telemedicine and point–of–care devices for diagnostics. Some of the noted advantages of using smartphones as diagnostic tools include:
- Readily available sensors such as cameras (CMOS⁄CCD), gyros, accelerometers, GPS, magnetic field sensing etc.
- Computational power for computational–based applications such as lens less imaging.
- Handheld, cost–effective, portable device for point–ofcare diagnostics in a limited–resource setting.
- Availability of advanced treatments to remote parts of the world.
- Networking of the data gathered, through wireless communication.
- Data uploaded to central servers wirelessly can be used to create spatio–temporal maps of various conditions visualized in real time. Thereby, realizing a distributed sensing approach which allows for application of smartphones for disease surveillance and management as well as epidemiologic studies in general.
- Most Smartphone–based devices consist of an accessory which could be attached to a smartphone. Such an arrangement allows for the use of different accessories to realize different diagnostic tools on a single cellular device.
Almost all of the smartphone–based devices that have been reported so far are realized through the attachment of an external module to smartphones. The external modules usually make use of the Smartphone’s camera. More importantly, it is the design of these external modules⁄attachments that determines the functionality of the smartphone–based device as a whole. Some of the external modules⁄ attachments for the Smartphone’s camera and their applications are discussed below:
External modules for Spectroscopy
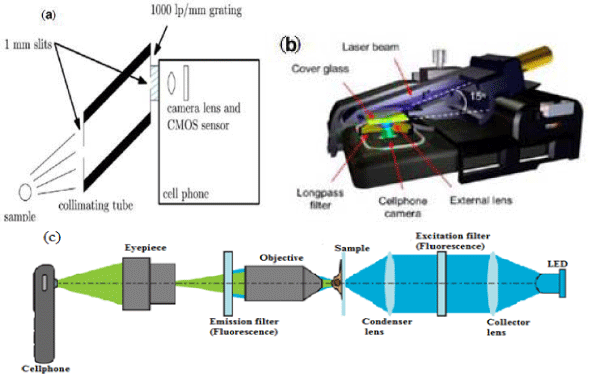
The main objective for this external module would be to decompose the incoming light into its spectral components. One such module proposed by Smith et al. [149] consists of a collimating tube with slits on both ends Figure 11(a). The first slit (the one closer to the sample) controls the resolution of the spectrometer. Better resolutions could be obtained by minimizing the aperture of the first slit. The second slit, in this case was fixed at 1 mm. The second slit forms the aperture for the diffraction grating where light is split into its respective components and is captured by the smartphone camera. The spectral components are then quantified from the captured image. Gallegos et al, [155] demonstrated the use of a smartphone spectrometer as the detection instrument for a label–free photonic crystal biosensor and has achieved 0.009nm accuracy in measuring shifts in the resonant wavelength of the sensor. Public Lab introduced a smartphone spectrometer kit at the price of $70 [156]. Wang et al. [148] has proposed different ways for implementing smartphonebased spectroscopy. A few of their designs for externals modules include techniques using rotating optical filter wheels, acoustooptical tunable filter (AOTF), liquid crystal tunable filter (LCTF), and diffraction grating. It is important to note that spectral images could be obtained by using filters such as rotating filter wheels.
Figure 11: (a)System diagram for a smartphone-based spectrometry [149]. (Permission to reuse to be obtained from PLOS ONE); (b) Ray-tracing diagram of the smartphone microscope is shown, where excitation and scattered beams are indicated with solid blue rays, while the fluorescent emission is highlighted with solid green rays [153]. (Permission to reuse to be obtained from ACS) (c) Smartphone-based microscopy, an optical layout for fluorescence imaging. The same apparatus can be used for bright field imaging, with the filters and LED removed [157]. (Permission to reuse to be obtained from PLOS ONE).
External module for Fluorescence imaging of single nanoparticle
Wei et al. [153] used a field–portable fluorescence microscopy platform installed on a smartphone for imaging of individual nanoparticles as well as viruses. The external module used in the platform houses a compact laser–diode–based excitation at 450 nm that illuminates the sample plane at a high incidence angle, a longpass (LP) thin film interference filter, an external low NA lens, and a coarse mechanical translation stage or focusing and depth adjustment Figure 11(b).The low NA (numerical aperture) external collection lens rejects the oblique illumination light on the sample plane. The scattered light from the sample plane is filtered by the LP filter, acting as a background rejection mechanism which is necessary to isolate the weak fluorescent signal arising from individual nanoparticles or viruses (on the sample plane). The low NA imaging system is also reduces the alignment sensitivity to depth of field, such that a coarse mechanical translation stage could be sufficient to focus the cell phone microscope to the sample plane.
External modules for Microscopy
One of proposed designs for smartphone–based microscopy is discussed here. Breslauer et al. [157] reported a system using a 0.85 NA 60×achromatic objective and a 20x wide–field microscope eyepiece. A schematic diagram of the system is as shown in Figure 11(c). The resulting system features a field–of–view of 180 mm diameter, an effective magnification of 28× onto the camera face and a measured spatial resolution of 1.2 mm. This system could be used both for bright field microscopy and fluoroscopy (using fluorescence filters as shown in the figure).
Many other external modules have also been reported in the literatures but only a few of them have been discussed here for conceptual purposes. Key applications such as spectroscopy, microscopy and fluorescence imaging have been demonstrated with smartphone–based devices which show the potential for costeffective, point–of–care diagnostic tool with wireless communication. Smartphone–based microscopy has been used for observing blood smears, imaging tuberculosis, imaging malaria and sickle–cell anemia [157]. Smartphone–based spectroscopy has been used to observe the transmission spectra from a finger [149]. However, current smartphone–based device accessories lack the integration of a fiberoptic attachment which would provide ease of access to cervical, oral and anal cancer sites. Advancements in both smartphones and external modules⁄attachments could provide a more robust device for diagnosis. Although, Wang et al. [148] mentioned a fiber–optic external module which could collect the diffuse reflectance, very limited information has been reported in the literatures about the use of an external fiber optic module with smart–phones.
In conclusion, smartphone–based devices show great potential for point–of–care diagnostic tool that is particularly useful for cervical, oral and anal cancer detection in developing countries. Realization of fiber–optic external modules needs to be explored which could open the doors for smartphone–based endoscopy and optical diagnostics.
Summary
Despite many improvements over the last decades, prevention, screening and diagnostic tests of cancers in the oropharynx and anogenital regions remain a huge challenge in developing countries and resource–poor areas. This is mainly due to the lack of appropriate medical infrastructure, expensive resources and well–trained personnel to support the organized screening and diagnostic programsthat are available in the developed world. Emerging optical diagnostic techniques, especially VIS–DRS, reflectance and fluorescence imaging as well as smartphone based devices, are noninvasive technologies that have shown great potential as a cost–effective and fast tool for diagnosis of early precancerous changes in the cervix, oral cavity and anus. Among these techniques, VIS–DRS with a fiber–optic probe is one of the few that can provide quantitative diagnosis about tissue optical and physiological properties. Portable fluorescence imaging systems is another promising candidate for addressing the current challenges. Smartphone based devices are likely to play an increasing role in the near future.
The high resolution and sensitivity offered by optical diagnostic techniques make them promising solutions to the health problems related to oral, cervical and anal cancers in resource poor settings. The cost consideration also favors optical techniques, as they can be manufactured with low cost. With available photonics technologies, miniature CCDs and spectrometers can now be manufactured and integrated into optical diagnostic systems at low cost, provided that there is enough need. Modular design is another effective approach to reduce the system complexity and associated cost. In fact, turning massively available spare smartphones into diagnostically useful optical imaging or spectroscopy devices has been a hot research topic of the recent Global Health programs at NIH as well as the Bill & Melinda Gates Foundation.
Despite of the encouraging progress in the past decade, optical diagnostic techniques for developing countries are still in their nascent research stage. More coordinated efforts from individual research groups as well as related industries are needed to move these techniques forward. In addition, the use of optical imaging and spectroscopy will be primarily compatible with assessable organs due to the limitation of light penetration in deeply embedded tissues.
In the next stage of development, it will be likely that the research community will establish a common calibration standard, which will enhance the quality control and clinical systems. From this point of view, the smart fiber–optic sensor device which includes a self–calibration and a pressure channel is an important step towards the goal of obtaining consistently calibrated and quantitative measurements that can be compared across different studies from different groups.
Larger scale clinical studies to compare sensitivity and specificity of optical techniques with that of the gold standard, such as pathology, are also needed to facilitate the clinical translation of various optical systems. The cost evaluation of optical techniques versus traditional smear test needs to be evaluated. In low–resource settings, the “valuedriven engineering” approach may help us better understand the advantages of disadvantages of new optical techniques.
References
- Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010; 127: 2893-2917.
- Vizcaino AP, Moreno V, Bosch FX, Muñoz N, Barros-Dios XM, Borras J, et al. International trends in incidence of cervical cancer: II. Squamous-cell carcinoma. Int J Cancer. 2000; 86: 429-435.
- Westra WH. The changing face of head and neck cancer in the 21st century: the impact of HPV on the epidemiology and pathology of oral cancer. Head Neck Pathol. 2009; 3: 78-81.
- Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005; 55: 74-108.
- Licitra L, Bernier J, Grandi C, Merlano M, Bruzzi P, Lefebvre JL. Cancer of the oropharynx. Crit Rev Oncol Hematol. 2002; 41: 107-122.
- Dahlstrand HM, Dalianis T. Presence and influence of human papillomaviruses (HPV) in Tonsillar cancer. Adv Cancer Res. 2005; 93: 59-89.
- Johnson LG, Madeleine MM, Newcomer LM, Schwartz SM, Daling JR. Anal cancer incidence and survival: the surveillance, epidemiology, and end results experience, 1973-2000. Cancer. 2004; 101: 281-288.
- Abbas A, Yang G, Fakih M. Management of anal cancer in 2010. Part 2: current treatment standards and future directions. Oncology (Williston Park). 2010; 24: 417-424.
- Bjørge T, Engeland A, Luostarinen T, Mork J, Gislefoss RE, Jellum E, et al. Human papillomavirus infection as a risk factor for anal and perianal skin cancer in a prospective study, Br J Cancer, 2002; 87: 61-64.
- Frisch M, Glimelius B, van den Brule AJ, Wohlfahrt J, Meijer CJ, Walboomers JM, et al. Sexually transmitted infection as a cause of anal cancer. N Engl J Med. 1997; 337: 1350-1358.
- Varnai AD, Bollmann M, Griefingholt H, Speich N, Schmitt C, Bollmann R, et al. HPV in anal squamous cell carcinoma and anal intraepithelial neoplasia (AIN). Impact of HPV analysis of anal lesions on diagnosis and prognosis, Int J Colorectal Dis. 2006; 21: 135-142.
- Abramowitz L, Jacquard AC, Jaroud F, Haesebaert J, Siproudhis L, Pradat P, et al. Human papillomavirus genotype distribution in anal cancer in France: the EDiTH V study, Int J Cancer. 2011; 129: 433-439.
- Hoots BE, Palefsky JM, Pimenta JM, Smith JS. Human papillomavirus type distribution in anal cancer and anal intraepithelial lesions. Int J Cancer. 2009; 124: 2375-2383..
- Wong AK, Chan RC, Aggarwal N, Singh MK, Nichols WS, Bose S. Human papillomavirus genotypes in anal intraepithelial neoplasia and anal carcinoma as detected in tissue biopsies, Mod Pathol. 2010; 23: 144-150.
- Green GE, Bauman NM, Smith RJ. Pathogenesis and treatment of juvenile onset recurrent respiratory papillomatosis. Otolaryngol Clin North Am. 2000; 33: 187-207.
- Ferris DG, Cox JT, O'Connor DM, Wright VC, Foerster J. Modern Colposcopy: Textbook and Atlas. 2nd edn. Kendal Hunt 2007.
- Villa LL, Costa RL, Petta CA, Andrade RP, Ault KA, Giuliano AR, et al. Prophylactic quadrivalent human papillomavirus (types 6, 1, 16, and 18) L1 virus-like particle vaccine in young women: a randomised double-blind placebo-controlled multicentre phase II efficacy trial, Lancet Oncol. 2005; 6: 271-278.
- Mao C, Koutsky LA, Ault KA, Wheeler CM, Brown DR, Wiley DJ, et al. Efficacy of human papillomavirus-16 vaccine to prevent cervical intraepithelial neoplasia: a randomized controlled trial, Obstet Gynecol. 2006; 107: 18-27.
- Harper DM, Franco EL, Wheeler CM, Moscicki AB, Romanowski B, Roteli-Martins CM, et al. Sustained efficacy up to 4.5 years of a bivalent L1 virus-like particle vaccine against human papillomavirus types 16 and 18: follow-up from a randomised control trial, Lancet. 2006; 367:1247-1255.
- Harper DM, Franco EL, Wheeler C, Ferris DG, Jenkins D, Schuind A, et al. Efficacy of a bivalent L1 virus-like particle vaccine in prevention of infection with human papillomavirus types 16 and 18 in young women: a randomised controlled trial, Lancet. 2004; 364: 1757-1765.
- Garland SM, Hernandez-Avila M, Wheeler CM, Perez G, Harper DM, Leodolter S, et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007; 356: 1928-1943.
- FUTURE II Study Group. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med. 2007; 356: 1915-1927.
- Ault KA. Future II Study Group. Effect of prophylactic human papillomavirus L1 virus-like-particle vaccine on risk of cervical intraepithelial neoplasia grade 2, grade 3, and adenocarcinoma in situ: a combined analysis of four randomised clinical trials. Lancet. 2007; 369: 1861-1868.
- HPV vaccine: Safe, effective, and grossly underutilized, Centers for Disease Control and Prevention. 2013.
- Brotherton JM, Fridman M, May CL, Chappell G, Saville AM, Gertig DM. Early effect of the HPV vaccination programme on cervical abnormalities in Victoria, Australia: an ecological study. Lancet. 2011; 377: 2085-2092.
- Ali H, Donovan B, Wand H, Read TR, Regan DG, Grulich AE, et al. Genital warts in young Australians five years into national human papillomavirus vaccination programme: national surveillance data. BMJ. 2013; 346: f2032.
- Ali H, Guy RJ, Wand H, Read TR, Regan DG, Grulich AE, et al. Decline in in-patient treatments of genital warts among young Australians following the national HPV vaccination program. BMC Infect Dis. 2013; 13: 140.
- Donovan B, Franklin N, Guy R, Grulich AE, Regan DG, Ali H, et al. Quadrivalent human papillomavirus vaccination and trends in genital warts in Australia: analysis of national sentinel surveillance data. Lancet Infect Dis. 2011; 11: 39-44.
- Goldstone S, Palefsky JM, Giuliano AR, Moreira ED Jr, Aranda C, Jessen H, et al. Prevalence of and risk factors for human papillomavirus (HPV) infection among HIV-seronegative men who have sex with men. J Infect Dis. 2011; 203: 66-74.
- Li AH, Phanuphak N, Sahasrabuddhe VV, Chaithongwongwatthana S, Vermund SH, Jenkins CA, et al. Anal squamous intraepithelial lesions among HIV positive and HIV negative men who have sex with men in Thailand. Sex Transm Infect. 2009; 85: 503-507.
- Panther LA, Wagner K, Proper J, Fugelso DK, Chatis PA, Weeden W, et al. High resolution anoscopy findings for men who have sex with men: inaccuracy of anal cytology as a predictor of histologic high-grade anal intraepithelial neoplasia and the impact of HIV serostatus. Clin Infect Dis. 2004; 38: 1490-1492.
- Koliopoulos G, Arbyn M, Martin-Hirsch P, Kyrgiou M, Prendiville W, Paraskevaidis E. Diagnostic accuracy of human papillomavirus testing in primary cervical screening: a systematic review and meta-analysis of non-randomized studies, Gynecol Oncol. 2007; 104: 232-246.
- Runowicz CD. Molecular screening for cervical cancer--time to give up Pap tests? N Engl J Med. 2007; 357: 1650-1653.
- Cuzick J, Szarewski A, Terry G, Ho L, Hanby A, Maddox P, et al. Human papillomavirus testing in primary cervical screening. Lancet. 1995; 345: 1533-1536.
- Denny L, Kuhn L, Pollack A, Wainwright H, Wright TC Jr. Evaluation of alternative methods of cervical cancer screening for resource-poor settings. Cancer. 2000; 89: 826-833.
- Schiffman M, Herrero R, Hildesheim A, Sherman ME, Bratti M, Wacholder S. HPV DNA testing in cervical cancer screening: results from women in a high-risk province of Costa Rica. JAMA. 2000; 283: 87-93.
- Kuhn L, Denny L, Pollack A, Lorincz A, Richart RM, Wright TC. Human papillomavirus DNA testing for cervical cancer screening in low-resource settings. J Natl Cancer Inst. 2000; 92: 818-825.
- Wright TC Jr, Massad LS, Dunton CJ, Spitzer M, Wilkinson EJ, Solomon D, et al. 2006 American Society for Colposcopy and Cervical Pathology-sponsored Consensus Conference. 2006 consensus guidelines for the management of women with cervical intraepithelial neoplasia or adenocarcinoma in situ. Am J Obstet Gynecol. 2007; 197: 340-345.
- Cuzick J, Arbyn M, Sankaranarayanan R, Tsu V, Ronco G, Mayrand MH, et al. Overview of human papillomavirus-based and other novel options for cervical cancer screening in developed and developing countries. Vaccine. 2008; 26 Suppl 10: K29-41.
- Zhao FH, Jeronimo J, Qiao YL, Schweizer J, Chen W, Valdez M, et al. An evaluation of novel, lower-cost molecular screening tests for human papillomavirus in rural China, Cancer Prev Res (Phila). 2013; 6: 938-948.
- Visual inspection with acetic acid for cervical-cancer screening: test qualities in a primary-care setting. University of Zimbabwe/JHPIEGO Cervical Cancer Project. Lancet. 1999; 353: 869-873.
- Megevand E, Denny L, Dehaeck K, Soeters R, Bloch B. Acetic acid visualization of the cervix: an alternative to cytologic screening. Obstet Gynecol. 1996; 88: 383-386.
- Sankaranarayanan R, Basu P, Wesley RS, Mahe C, Keita N, Mbalawa CC, et al. Accuracy of visual screening for cervical neoplasia: Results from an IARC multicentre study in India and Africa. Int J Cancer. 2004; 110: 907-913.
- Sankaranarayanan R, Wesley R, Thara S, Dhakad N, Chandralekha B, Sebastian P, et al. Test characteristics of visual inspection with 4% acetic acid (VIA) and Lugol's iodine (VILI) in cervical cancer screening in Kerala, India. Int J Cancer. 2003; 106: 404-408.
- Sankaranarayanan R, Nene BM, Shastri SS, Jayant K, Muwonge R, Budukh AM et al HPV screening for cervical cancer in rural India. N Engl J Med. 2009; 360: 1385-1394.
- Goldie SJ, Gaffikin L, Goldhaber-Fiebert JD, Gordillo-Tobar A, Levin C, et al. Cost-effectiveness of cervical-cancer screening in five developing countries, N Engl J Med. 2005; 353: 2158-2168.
- Ferris DG, Litaker MS; ASCUS/LSIL Triage Study (ALTS) Group. Colposcopy quality control by remote review of digitized colposcopic images. Am J Obstet Gynecol. 2004; 191: 1934-1941.
- Massad LS, Jeronimo J, Katki HA, Schiffman M. National Institutes of Health/American Society for Colposcopy and Cervical Pathology Research Group. The accuracy of colposcopic grading for detection of high-grade cervical intraepithelial neoplasia. J Low Genit Tract Dis. 2009; 13: 137-144.
- Hammes LS, Naud P, Passos EP, Matos J, Brouwers K, Rivoire W, et al. Value of the International Federation for Cervical Pathology and Colposcopy (IFCPC) Terminology in predicting cervical disease. J Low Genit Tract Dis. 2007; 11: 158-165.
- Cantor SB, Cárdenas-Turanzas M, Cox DD, Atkinson EN, Nogueras-Gonzalez GM, Beck JR, et al. Accuracy of colposcopy in the diagnostic setting compared with the screening setting. Obstet Gynecol. 2008; 111: 7-14.
- Moss EL, Dhar KK, Byrom J, Jones PW, Redman CW. The diagnostic accuracy of colposcopy in previously treated cervical intraepithelial neoplasia. J Low Genit Tract Dis. 2009; 13: 5-9.
- Wentzensen N, Zuna RE, Sherman ME, Gold MA, Schiffman M, Dunn ST, et al. Accuracy of cervical specimens obtained for biomarker studies in women with CIN3. Gynecol Oncol. 2009; 115: 493-496.
- Pretorius RG, Zhang WH, Belinson JL, Huang MN, Wu LY, Zhang X, et al. Colposcopically directed biopsy, random cervical biopsy, and endocervical curettage in the diagnosis of cervical intraepithelial neoplasia II or worse, Am J Obstet Gynecol. 2004; 191: 430-434.
- Stoler MH, Vichnin MD, Ferenczy A, Ferris DG, Perez G, Paavonen J, et al. The accuracy of colposcopic biopsy: analyses from the placebo arm of the Gardasil clinical trials. Int J Cancer. 2011; 128: 1354-1362.
- Jeronimo J, Schiffman M. Colposcopy at a crossroads. Am J Obstet Gynecol. 2006; 195: 349-353.
- Gage JC, Hanson VW, Abbey K, Dippery S, Gardner S, Kubota J, et al. Number of cervical biopsies and sensitivity of colposcopy. Obstet Gynecol. 2006; 108: 264-272.
- Hakama M, Magnus K, Pettersson F, Storm H, Tulinius H. Effect of organized screening on the risk of cervical cancer in Nordic countries, in Cancer Screening, Miller A, et al., editors, pp 153-162 Cambridge University Press: Cambridge. 1991.
- SEER Program, National Cancer Institute, USA. 1999.
- Koss LG. The Papanicolaou test for cervical cancer detection. A triumph and a tragedy. JAMA. 1989; 261: 737-743.
- Ferris DG, Berrey MM, Ellis KE, Petry LJ, Voxnaes J, Beatie RT. The optimal technique for obtaining a Papanicolaou smear with the Cervex-Brush. J Fam Pract. 1992; 34: 276-280.
- Cox JT. Management of cervical intraepithelial neoplasia. Lancet. 1999; 353: 857-859.
- Richart RM, Wright TC Jr. Controversies in the management of low-grade cervical intraepithelial neoplasia. Cancer. 1993; 71: 1413-1421.
- Centers for Disease Control (CDC). Cervical cancer control--Rhode Island. MMWR Morb Mortal Wkly Rep. 1989; 38: 659-662.
- Friedell GH, Tucker TC, McManmon E, Moser M, Hernandez C, Nadel M. Incidence of dysplasia and carcinoma of the uterine cervix in an Appalachian population. J Natl Cancer Inst. 1992; 84: 1030-1032.
- Wied GL, Bartels PH, Bibbo M, Keebler CM. Frequency and reliability of diagnostic cytology of the female genital tract. Acta Cytol. 1981; 25: 543-549.
- A.f.H.C.P.a. Research, Evidence report/technology assessment No 5: evaluation of cervical cytology, Rockville, MD: Agency for Health Care Policy and Research, AHCPR publication No.99-E010, U.S. Department of Health and Human Services. 1999.
- Giles JA, Hudson E, Crow J, Williams D, Walker P. Colposcopic assessment of the accuracy of cervical cytology screening. Br Med J (Clin Res Ed). 1988; 296: 1099-1102.
- Stoler MH, Schiffman M; Atypical Squamous Cells of Undetermined Significance-Low-grade Squamous Intraepithelial Lesion Triage Study (ALTS) Group. Interobserver reproducibility of cervical cytologic and histologic interpretations: realistic estimates from the ASCUS-LSIL Triage Study. JAMA. 2001; 285: 1500-1505.
- Paz Soldan VA, Lee FH, Carcamo C, Holmes KK, Garnett GP, Garcia P. Who is getting Pap smears in urban Peru? Int J Epidemiol. 2008; 37: 862-869.
- Huang D, Swanson EA, Lin CP, Schuman JS, Stinson WG, Chang W, et al. Optical coherence tomography. Science. 1991; 254: 1178-1181.
- Lee CK, Chi TT, Wu CT, Tsai MT, Chiang CP, Yang CC, et al. Diagnosis of oral precancer with optical coherence tomography. Biomed Opt Express. 2012; 3: 1632-1646.
- Ovári A, Pau HW, Just T. [Optical coherence tomography in otolaryngology]. Orv Hetil. 2011; 152: 1125-1132.
- Liu Z, Belinson SE, Li J, Yang B, Wulan N, Tresser NJ, et al. Diagnostic efficacy of real-time optical coherence tomography in the management of preinvasive and invasive neoplasia of the uterine cervix, Int J Gynecol Cancer. 2010; 20: 283-287.
- Gallwas JK, Turk L, Stepp H, Mueller S, Ochsenkuehn R, Friese K, et al. Optical coherence tomography for the diagnosis of cervical intraepithelial neoplasia. Lasers Surg Med. 2011; 43: 206-212.
- Zhang QQ, Wu XJ, Tang T, Zhu SW, Yao Q, Gao BZ, et al. Quantitative analysis of rectal cancer by spectral domain optical coherence tomography. Phys Med Biol. 2012; 57: 5235-5244.
- Keller DM. Optical Approaches to Oral Cancer Screening and Diagnosis: An Expert Interview With Petra Wilder-Smith DDS, PhD , Medscape. 2010.
- Clark AL, Gillenwater AM, Collier TG, Alizadeh-Naderi R, El-Naggar AK, Richards-Kortum RR. Confocal microscopy for real-time detection of oral cavity neoplasia. Clin Cancer Res. 2003; 9: 4714-4721.
- Carlson K, Pavlova I, Collier T, Descour M, Follen M, Richards-Kortum R. Confocal microscopy: imaging cervical precancerous lesions. Gynecol Oncol. 2005; 99: S84-88.
- Youmin Wang, Milan Raj, Stan McGuff H, Gauri Bhave, Bin Yang, Ting Shen, et al. Portable oral cancer detection using a miniature confocal imaging probe with a large field of view, J Micromech. Microeng. 2012; 22: 065001.
- Roblyer D, Richards-Kortum R, Sokolov K, El-Naggar AK, Williams MD, Kurachi C. Multispectral optical imaging device for in vivo detection of oral neoplasia. J Biomed Opt. 2008; 13: 024019.
- Noninvasive Optical System Reveals Oral Cancer Signs, Photonics.com, KERALA, India. 2014.
- Zonios G, Perelman LT, Backman V, Manoharan R, Fitzmaurice M, Van Dam J, et al. Diffuse reflectance spectroscopy of human adenomatous colon polyps in vivo. Appl Opt. 1999; 38: 6628-6637.
- Murphy BW, Webster RJ, Turlach BA, Quirk CJ, Clay CD, Heenan PJ, et al. Toward the discrimination of early melanoma from common and dysplastic nevus using fiber optic diffuse reflectance spectroscopy. J Biomed Opt. 2005; 10: 064020.
- Randeberg LL. in Department of Electronics and Telecommunications. Norwegian University of Science and Technology. 2005.
- Yu B, Fu H, Bydlon T, Bender JE, Ramanujam N. Diffuse reflectance spectroscopy with a self-calibrating fiber optic probe. Opt Lett. 2008; 33: 1783-1785.
- Alerstam E, Andersson-Engels S, Svensson T. Improved accuracy in time-resolved diffuse reflectance spectroscopy. Opt Express. 2008; 16: 10440-10454.
- Mourant JR, Bigio IJ, Boyer J, Conn RL, Johnson T, Shimada T. Spectroscopic diagnosis of bladder cancer with elastic light scattering. Lasers Surg Med. 1995; 17: 350-357.
- Bigio IJ, Mourant JR, Los G. Noninvasive, in-situ measurement of drug concentrations in tissue using optical spectroscopy. J Gravit Physiol. 1999; 6: P173-175.
- Bigio IJ, Bown SG, Briggs G, Kelley C, Lakhani S, Pickard D, et al. Diagnosis of breast cancer using elastic-scattering spectroscopy: preliminary clinical results, J Biomed Opt. 2000; 5: 221-228.
- Ntziachristos V, Yodh AG, Schnall MD, Chance B. MRI-guided diffuse optical spectroscopy of malignant and benign breast lesions. Neoplasia. 2002; 4: 347-354.
- Shah N, Cerussi AE, Jakubowski D, Hsiang D, Butler J, Tromberg BJ. The role of diffuse optical spectroscopy in the clinical management of breast cancer. Dis Markers. 2003; 19: 95-105.
- Cerussi A, Shah N, Hsiang D, Durkin A, Butler J, Tromberg BJ. In vivo absorption, scattering, and physiologic properties of 58 malignant breast tumors determined by broadband diffuse optical spectroscopy, J Biomed Opt. 2006; 11: 044005.
- Farrell TJ, Patterson MS, Wilson B. A diffusion theory model of spatially resolved, steady-state diffuse reflectance for the noninvasive determination of tissue optical properties in vivo, Med Phys. 1992; 19: 79-88.
- Volynskaya Z, Haka AS, Bechtel KL, Fitzmaurice M, Shenk R, Wang N, et al. Diagnosing breast cancer using diffuse reflectance spectroscopy and intrinsic fluorescence spectroscopy, J Biomed Opt. 2008; 13: 024012.
- Palmer GM, Ramanujam N. Monte Carlo-based inverse model for calculating tissue optical properties. Part I: Theory and validation on synthetic phantoms. Appl Opt. 2006; 45: 1062-1071.
- Arifler D, Schwarz RA, Chang SK, Richards-Kortum R. Reflectance spectroscopy for diagnosis of epithelial precancer: model-based analysis of fiber-optic probe designs to resolve spectral information from epithelium and stroma, Appl Opt. 2005; 44: 4291-4305.
- Thueler P, Charvet I, Bevilacqua F, St Ghislain M, Ory G, Marquet P, et al. In vivo endoscopic tissue diagnostics based on spectroscopic absorption, scattering, and phase function properties, J Biomed Opt. 2003; 8: 495-503.
- Wang Q, Yang H, Agrawal A, Wang NS, Pfefer TJ. Measurement of internal tissue optical properties at ultraviolet and visible wavelengths: Development and implementation of a fiberoptic-based system. Opt Express. 2008; 16: 8685-8703.
- Reif R, A'Amar O, Bigio IJ. Analytical model of light reflectance for extraction of the optical properties in small volumes of turbid media. Appl Opt. 2007; 46: 7317-7328.
- Yu B, Shah A, Nagarajan VK, Ferris DG. Diffuse reflectance spectroscopy of epithelial tissue with a smart fiber-optic probe. Biomed Opt Express. 2014; 5: 675-689.
- Skala MC, Palmer GM, Vrotsos KM, Gendron-Fitzpatrick A, Ramanujam N. Comparison of a physical model and principal component analysis for the diagnosis of epithelial neoplasias in vivo using diffuse reflectance spectroscopy, Opt Express. 2007; 15: 7863-7875.
- Chang VT, Cartwright PS, Bean SM, Palmer GM, Bentley RC, Ramanujam N. Quantitative physiology of the precancerous cervix in vivo through optical spectroscopy. Neoplasia. 2009; 11: 325-332.
- Müller MG, Valdez TA, Georgakoudi I, Backman V, Fuentes C, Kabani S, et al. Spectroscopic detection and evaluation of morphologic and biochemical changes in early human oral carcinoma. Cancer. 2003; 97: 1681-1692.
- de Veld DC, Skurichina M, Witjes MJ, Duin RP, Sterenborg HJ, Roodenburg JL. Autofluorescence and diffuse reflectance spectroscopy for oral oncology. Lasers Surg Med. 2005; 36: 356-364.
- Sharwani A, Jerjes W, Salih V, Swinson B, Bigio IJ, El-Maaytah M, et al. Assessment of oral premalignancy using elastic scattering spectroscopy. Oral Oncol. 2006; 42: 343-349.
- Subhash N, Mallia JR, Thomas SS, Mathews A, Sebastian P, Madhavan J. Oral cancer detection using diffuse reflectance spectral ratio R540/R575 of oxygenated hemoglobin bands. J Biomed Opt. 2006; 11: 014018.
- Amelink A, Kaspers OP, Sterenborg HJ, van der Wal JE, Roodenburg JL, Witjes MJ. Non-invasive measurement of the morphology and physiology of oral mucosa by use of optical spectroscopy. Oral Oncol. 2008; 44: 65-71.
- Rahman M, Chaturvedi P, Gillenwater AM, Richards-Kortum R. Low-cost, multimodal, portable screening system for early detection of oral cancer. J Biomed Opt. 2008; 13: 030502.
- Schwarz RA, Gao W, Daye D, Williams MD, Richards-Kortum R, Gillenwater AM. Autofluorescence and diffuse reflectance spectroscopy of oral epithelial tissue using a depth-sensitive fiber-optic probe. Appl Opt. 2008; 47: 825-834.
- Mirabal YN, Chang SK, Atkinson EN, Malpica A, Follen M, Richards-Kortum R. Reflectance spectroscopy for in vivo detection of cervical precancer. J Biomed Opt. 2002; 7: 587-594.
- Chang SK, Mirabal YN, Atkinson EN, Cox D, Malpica A, Follen M, et al. Combined reflectance and fluorescence spectroscopy for in vivo detection of cervical pre-cancer. J Biomed Opt. 2005; 10: 024031.
- Follen M, Crain S, MacAulay C, Basen-Engquist K, Cantor SB, Cox D, et al. Optical technologies for cervical neoplasia: update of an NCI program project grant. Clin Adv Hematol Oncol. 2005; 3: 41-53.
- Marín NM, Milbourne A, Rhodes H, Ehlen T, Miller D, Benedet L, et al. Diffuse reflectance patterns in cervical spectroscopy. Gynecol Oncol. 2005; 99: S116-120.
- Arifler D, MacAulay C, Follen M, Richards-Kortum R. Spatially resolved reflectance spectroscopy for diagnosis of cervical precancer: Monte Carlo modeling and comparison to clinical measurements. J Biomed Opt. 2006; 11: 064027.
- Cardenas-Turanzas M, Freeberg JA, Benedet JL, Atkinson EN, Cox DD, Richards-Kortum R, et al. The clinical effectiveness of optical spectroscopy for the in vivo diagnosis of cervical intraepithelial neoplasia: where are we? Gynecol Oncol. 2007; 107: S138-146.
- Freeberg JA, Benedet JL, MacAulay C, West LA, Follen M. The performance of fluorescence and reflectance spectroscopy for the in vivo diagnosis of cervical neoplasia; point probe versus multispectral approaches, Gynecol Oncol. 2007; 107: S248-255.
- Wang A, Nammalavar V, Drezek R. Targeting spectral signatures of progressively dysplastic stratified epithelia using angularly variable fiber geometry in reflectance Monte Carlo simulations, J Biomed Opt. 2007; 12: 044012.
- Lane PM, Gilhuly T, Whitehead P, Zeng H, Poh CF, Ng S, et al. Simple device for the direct visualization of oral-cavity tissue fluorescence. J Biomed Opt. 2006; 11: 024006.
- Pavlova I, Weber CR, Schwarz RA, Williams MD, Gillenwater AM, Richards-Kortum R. Fluorescence spectroscopy of oral tissue: Monte Carlo modeling with site-specific tissue properties. J Biomed Opt. 2009; 14: 014009.
- Hasina R, Lingen MW. Angiogenesis in oral cancer. J Dent Educ. 2001; 65: 1282-1290.
- Georgakoudi I, Sheets EE, Müller MG, Backman V, Crum CP, Badizadegan K, et al. Trimodal spectroscopy for the detection and characterization of cervical precancers in vivo. Am J Obstet Gynecol. 2002; 186: 374-382.
- Liu Q, Ramanujam N. Sequential estimation of optical properties of a two-layered epithelial tissue model from depth-resolved ultraviolet-visible diffuse reflectance spectra. Appl Opt. 2006; 45: 4776-4790.
- Utzinger U, Richards-Kortum RR. Fiber optic probes for biomedical optical spectroscopy. J Biomed Opt. 2003; 8: 121-147.
- Reif R, Amorosino MS, Calabro KW, A'Amar O, Singh SK, Bigio IJ. Analysis of changes in reflectance measurements on biological tissues subjected to different probe pressures. J Biomed Opt. 2008; 13: 010502.
- Ti Y, Lin WC. Effects of probe contact pressure on in vivo optical spectroscopy. Opt Express. 2008; 16: 4250-4262.
- Nath A, Rivoire K, Chang S, Cox D, Atkinson EN, Follen M, et al. Effect of probe pressure on cervical fluorescence spectroscopy measurements. J Biomed Opt. 2004; 9: 523-533.
- Rivoire K, Nath A, Cox D, Atkinson EN, Richards-Kortum R, Follen M. The effects of repeated spectroscopic pressure measurements on fluorescence intensity in the cervix. Am J Obstet Gynecol. 2004; 191: 1606-1617.
- #128. Shangguan H, Prahl SA, Jacques SL, Casperson LW. Pressure effects on soft tissues monitored by changes in tissue, Proc SPIE. 1998; 3254.
- Atencio JAD, Guillén EEO, Montiel SVy, Rodríguez MC, Ramos JC, Gutiérrez JL, et al. Influence of probe pressure on human skin diffuse reflectance spectroscopy measurements, Optical Memory & Neural Networks. 2009; 18: 6-14.
- Chen W, Liu R, Xu K, Wang RK. Influence of contact state on NIR diffuse reflectance spectroscopy in vivo, J. Phys. D: Appl. Phys. 2005; 38: 2691–2695.
- Ruderman S, Gomes AJ, Stoyneva V, Rogers JD, Fought AJ, Jovanovic BD, et al. Analysis of pressure, angle and temporal effects on tissue optical properties from polarization-gated spectroscopic probe measurements, Biomed Opt Express. 2010; 1: 489-499.
- Chan EK, Sorg B, Protsenko D, O'Neil M, Motamedi M, Welch AJ. Effects of compression on soft tissue optical properties, Selected Topics in Quantum Electronics, IEEE Journal of. 1996; 2: 943- 950.
- Yu B, Chang V, Shah A, Merisier D, Walmer D, Ramanujam N. A smart fiber optic sensor for detection of oral and cervical cancer in developing world, NIH/NCI Cancer Detection and Diagnostics Technologies for Global Health Conference, NIH Campus, MD. 2011; 22-23.
- Drezek R, Brookner C, Pavlova I, Boiko I, Malpica A, Lotan R, et al. Autofluorescence microscopy of fresh cervical-tissue sections reveals alterations in tissue biochemistry with dysplasia. Photochem Photobiol. 2001; 73: 636-641.
- Pavlova I, Sokolov K, Drezek R, Malpica A, Follen M, Richards-Kortum R. Microanatomical and biochemical origins of normal and precancerous cervical autofluorescence using laser-scanning fluorescence confocal microscopy. Photochem Photobiol. 2003; 77: 550-555.
- Crane LM, Themelis G, Pleijhuis RG, Harlaar NJ, Sarantopoulos A, Arts HJ, et al. Intraoperative multispectral fluorescence imaging for the detection of the sentinel lymph node in cervical cancer: a novel concept. Mol Imaging Biol. 2011; 13: 1043-1049.
- Pavlova I, Williams M, El-Naggar A, Richards-Kortum R, Gillenwater A. Understanding the biological basis of autofluorescence imaging for oral cancer detection: high-resolution fluorescence microscopy in viable tissue. Clin Cancer Res. 2008; 14: 2396-2404.
- Poh CF, Ng SP, Williams PM, Zhang L, Laronde DM, Lane P, et al. Direct fluorescence visualization of clinically occult high-risk oral premalignant disease using a simple hand-held device. Head Neck. 2007; 29: 71-76.
- Shin D, Vigneswaran N, Gillenwater A, Richards-Kortum R. Advances in fluorescence imaging techniques to detect oral cancer and its precursors. Future Oncol. 2010; 6: 1143-1154.
- Thekkek N, Richards-Kortum R. Optical imaging for cervical cancer detection: solutions for a continuing global problem. Nat Rev Cancer. 2008; 8: 725-731.
- Pierce M, Yu D, Richards-Kortum R. High-resolution fiber-optic microendoscopy for in situ cellular imaging. J Vis Exp. 2011;.
- Pierce MC, Guan Y, Quinn MK, Zhang X, Zhang WH, Qiao YL, et al. A pilot study of low-cost, high-resolution microendoscopy as a tool for identifying women with cervical precancer. Cancer Prev Res (Phila). 2012; 5: 1273-1279.
- Liu Y, Bauer AQ, Akers WJ, Sudlow G, Liang K, Shen D, et al. Hands-free, wireless goggles for near-infrared fluorescence and real-time image-guided surgery. Surgery. 2011; 149: 689-698.
- Liu Y, Njuguna R, Matthews T, Akers WJ, Sudlow GP, Mondal S, et al. Near-infrared fluorescence goggle system with complementary metal-oxide-semiconductor imaging sensor and see-through display. J Biomed Opt. 2013; 18: 101303.
- Liu Y, Akers WJ, Bauer AQ, Mondal S, Gullicksrud K, Sudlow GP, et al. Intraoperative detection of liver tumors aided by a fluorescence goggle system and multimodal imaging. Analyst. 2013; 138: 2254-2257.
- Liu Y, Zhao YM, Akers W, Tang ZY, Fan J, Sun HC, et al. First in-human intraoperative imaging of HCC using the fluorescence goggle system and transarterial delivery of near-infrared fluorescent imaging agent: a pilot study, Transl Res. 2013; 162: 324-331.
- Zhang J. Spectrometers miniaturized for working with cellular phones and other portable electronic devices, Int. Patent Application No. PCT/US2008/083613. 2008.
- Wang S, Zhou X. Spectroscopic sensor on mobile phone, Patent No. US7420663. 2008.
- Chu K, Smith ZJ, Dwyre D, Matthews D, Lane S, Wachsmann-Hogiu S. Microscopy and Spectroscopy on a Cell Phone, Bio-Optics: Design and Application, Monterey, California United States, April 4-6, 2011.
- Iqbal Z, Eriksson M. Classification and quantitative optical analysis of liquid and solid samples using a mobile phone as illumination source and detector, Sensors and Actuators B: Chemical. 2013; 185: 354–362.
- Sumriddetchkajorn S, Somboonkaew A, Chanhorm S. Mobile device-based digital microscopy for education, healthcare, and agriculture, 9th International Conference on Electrical Engineering/Electronics, Computer, Telecommunications and Information Technology (ECTI-CON), Phetchaburi. May 16-18, 2012.
- Ayas S, Cupallari A, Ekiz OO, Kaya Y, Dana A. Counting Molecules with a Mobile Phone Camera Using Plasmonic Enhancement, ACS Photonics. 2014; 1: 17-26.
- Wei Q, Qi H, Luo W, Tseng D, Ki SJ, Wan Z, et al. Fluorescent imaging of single nanoparticles and viruses on a smart phone. ACS Nano. 2013; 7: 9147-9155.
- Tseng D, Mudanyali O, Oztoprak C, Isikman SO, Sencan I, Yaglidere O, et al. Lensfree microscopy on a cellphone. Lab Chip. 2010; 10: 1787-1792.
- Gallegos D, Long KD, Yu H, Clark PP, Lin Y, George S, et al. Label-free biodetection using a smartphone. Lab Chip. 2013; 13: 2124-2132.
- Colaner S. Public Labs Open Source Smartphone Spectrometer Let's You Find Your Wavelength, HotHardware, November 18, 2013.
- Breslauer DN, Maamari RN, Switz NA, Lam WA, Fletcher DA. Mobile phone based clinical microscopy for global health applications. PLoS One. 2009; 4: e6320.