
Research Article
Austin J Dermatolog. 2024; 11(1): 1111.
Vitiligo and Vitamin D Deficiency: A Case-Control Study
Abeer Albazali, PhD¹; Ashok Sharma, PhD²; Fatmah Altaweel, MD³*; Adnan Ahmad, MD³; Yasmeen Mandani, MD³; Mohammed Aladwani, MD³; Deemah Aldaihani, MD³
¹Head of Dermatology Department in Farwaniya Hospital, Kuwait
²Head of Vitiligo clinic in Dermatology department, Farwaniya Hospital, Kuwait
³Doctor in Dermatology department, Farwaniya Hospital, Kuwait
*Corresponding author: Fatmah Altaweel Doctor in Dermatology department, Farwaniya Hospital, Kuwait. Email: [email protected]
Received: December 26, 2023 Accepted: January 27, 2024 Published: February 03, 2024
Abstract
Background: Vitamin D is a fat-soluble vitamin that is synthesized in the skin under the influence of Ultraviolet (UV) light from the sun and its deficiency has been linked with increased risk of autoimmune disease onset and/or relapse. Vitiligo is a chronic autoimmune disease that alters melanocytes and stimulates the immune system’s cells.
Aim: To assess the correlation between vitamin D deficiency in patients affected with vitiligo in Farwaniya Hospital’s dermatology department, Kuwait.
Materials & Methods: This was a record base case control study that included both male and female patients diagnosed with vitiligo and above 18 years of age. We designated 19 patients who were diagnosed with vitiligo in the vitiligo group and 19 patients with other dermatological diseases in the control group. Both groups have serum vitamin D levels documented in the hospital’s system. We collected the data from Farwaniya Hospital patient’s database of vitiligo patients who attended in the year 2022.
Conclusion: According to our research, vitamin D levels in the case group were slightly lower than the control group, but no significant difference was observed.
Introduction
Vitiligo is a chronic autoimmune disease, in which the pathogenesis begins with altered melanocytes that stimulates the immune system’s cells. This triggers these cells to target melanocytes in the epidermis to be destroyed. Vitiligo can affect both genders and all skin types. It is characterized by depigmented skin macules. Vitiligo is more common in people with a positive family history of the same disorder or who have other autoimmune diseases such as: thyroid disease, Addison’s disease, psoriasis, rheumatoid arthritis, systemic lupus erythematosus, type 1 diabetes mellitus and pernicious anemia [1]. Vitamin D is a fat-soluble vitamin which is synthesized in the skin under the influence of UV light from the sun and it has a role in absorbing and regulating the serum levels of both calcium and phosphorus. It goes through several steps of metabolism that begins in the form of (25-hydroxyvitamin D) before finally converting into the hormonal form which is (1,25-dihydroxyvitamin D3) [2]. With regards to the innate immune system, vitamin D deficiency has been linked with an increase in the expression of nuclear-factor kappa B (NF-κB), which leads to an elevated levels of the main pro-inflammatory cytokines, such as interleukin-6 (IL-6) and monocyte chemoattractant protein-1 (MCP-1) [3]. Serum concentration of 25-OH D (total vitamin D) is considered to be the most reliable measure of overall vitamin D status and thus can be used to determine whether a patient is Vitamin D deficient [4]. The results are categorized according to the Ministry of Health in Kuwait’s Main Laboratory ranges, that is deficient if below 50 nmol/L, insufficient if 50-75 nmol/L, sufficient 75-125 nmol/L, ideal 125-175 nmol/L and toxic if above 375 nmol/L [5-8]. A study was conducted in 2012 in Saud University, Riyadh, Saudi Arabia which showed that vitamin D supplementation for patients with vitiligo while they are receiving phototherapy or corticosteroids proved to be successful in some patients. In vitro studies showed that vitamin D3 lead to an increase in tyrosinase enzyme activity in cultured human melanocytes. Also, in vivo studies showed that the application of topical 100 micrograms of cholecalciferol to the skin of mice helped to upregulate the numbers of L-3,4-dihydroxyphenylalanine-positive (DOPA-positive) melanocytes and helped increase the therapeutic effect of phototherapy which led to more cosmetically appealing results and a less duration of treatment [9].
A cross-sectional case-control study was conducted in Basrah, Iraq from November 2019 till December 2020 during which they noticed there is a strong link between vitiligo and vitamin D deficiency. Female patients with vitiligo and low serum vitamin D concentrations were more common than male patients affected with vitiligo. Moreover, they found that most patients with vitiligo were from the young population [10].
In an analytical cross-sectional descriptive study which was done in 2012 in the dermatology clinics of Qazvin, Department of Dermatology, Qazvin University of Medical Sciences, Qazvin, Iran. A sample of 100 patients with vitiligo were studied and it was observed that there was no difference in the serum vitamin D levels between males and females. Furthermore, patients with vitiligo who participated in the study had a difference in their serum vitamin D levels based on their exposure to the sun which may be an important factor for vitiligo patients [11].
Another cross-sectional study was conducted in Jordan, Department of Dermatology, Faculty of Medicine, Jordan University of Science and Technology in Irbid, Jordan. Where a sample of 100 patients diagnosed with vitiligo were collected, in addition to 100 patients in the control group from May 2018 to December 2018. They observed that vitamin D levels were low in both vitiligo and control groups of this population. Also, they found out that the disease is more common in females when compared to males [12].
A study was conducted in 2021 in the Department of epidemiology, School of Public Health, Zhejiang Chinese Medical University, Hangzhou, China, in the form of meta-analysis and two-sample Mendelian Randomization (MR) which showed no solid evidence for a direct causality between the levels of both 25-hydroxyvitamin D or 25-hydroxyvitamin D3 and the risk of developing vitiligo. Moreover, there was an inverse relationship between vitiligo and the serum levels of 25(OH) D based on meta-analysis of observational studies, but MR did not show an association. With regards to 25(OH)D3, meta-analysis and MR study did not reveal any proof of a relationship between the serum levels of 25(OH)D3 and the risk of vitiligo. In conclusion, there was no evidence that vitamin D supplementation can prevent vitiligo [13].
Another study in the form of a systematic review and meta-analysis was conducted in 2020 in India which revealed that serum levels of vitamin D is lower in the vitiligo patients group when compared to the control group. Additionally, the study showed that vitamin D receptor polymorphism are linked with vitamin D levels and show a genetic predisposition for vitiligo [4].
According to a study which was conducted by Elio Kechichian et al. in 2017, vitamin D deficiency is associated with many dermatological diseases such as vitiligo, atopic dermatitis, psoriasis, alopecia areata, and polymorphic light eruption. For vitiligo, it was not clear if vitamin D deficiency was linked with the pathogenesis of vitiligo [15].
Materials and Methods
Case control study was performed. Data collection using a questionnaire from Farwaniya Hospital health record HIS (Health Information System). Demographic and clinical features were collected through the records, and these features include age, gender and nationality, duration & distribution of vitiligo lesion, medication patient was receiving and comorbidities. The sample size was all vitiligo patients of age 18 years and above who have attended Farwaniya hospital's dermatology department between January 2022 to December 2022 and have had a vitamin D blood test prior to receiving treatment. Eligible patients are men and women diagnosed to have vitiligo by a dermatologist. The exclusion criteria were as follows; participants less than 18 years of age, patients with vitiligo that are on vitamin D supplements, patients with vitiligo who received vitamin D supplements in the past three months, patients diagnosed with diabetes mellitus, thyroid disease, pregnant and lactating women [1]. Questionnaire was filled by the research team as per attached [appendice 1]. A group of 19 patients not related to the patients and fulfilled the inclusion and exclusion criteria were randomly picked from HIS as our control group.
Blood samples were viewed from both patients and controls to check vitamin D levels. The interpretation of the value of vitamin D was performed by a dermatologist and the subjects are classified based on MOH classification that is: deficient if below 50 nmol/L, insufficient if 50-75 nmol/L, sufficient 75-125 nmol/L, ideal 125-175 nmol/L and toxic if above 375 nmol/L [5-8].
Approval of research and ethical committee in Dermatology department in Farwaniya Hospital. Approval of Farwaniya Hospital administration center. Approval of ministry of health in Kuwait with a code of 2359-2023. Privacy and confidentiality of the recorded information is essential.
Statistical Methodology: Data were collected and coded then entered an IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp. Entered data for age and vitamin D levels were checked for accuracy then for normality, using Kolmogorov-Smirnov & Shapiro-Wilk tests, and proved to be not normally distributed. Thus, qualitative variables were expressed as number and percentage while quantitative variables were expressed as median and interquartile range (Q1-Q3). The median was used as measure of central tendency, while the interquartile range was used as a measure of dispersion.
The following statistical tests were used: independent samples Mann-Whitney’s U-test (or Z-test) was used for age and vitamin D level as a nonparametric test of significance for comparison between two sample medians; the X2-test (or likelihood ratio=LLR) was used as a non-parametric test of significance for comparison between the distribution of two qualitative variables; the Fisher’s exact test was used as a non-parametric test of significance for comparison between the distribution of two qualitative variables whenever the X2-test was not appropriate. It gives a p-value directly. A five percent level is chosen as a level of significance in all statistical significance tests used.
Results
A sample of 65 patients with vitiligo has been collected for the time between January 2022 to December 2022. 22 cases have been excluded due to age, 19 cases had missing data, so they were dismissed, and five cases were excluded as they were not fitting the inclusion criteria in terms of chronic diseases. 19 patients with vitiligo were recorded and approved to complete the research. In this study we have observed that there was almost no difference in age and gender between vitiligo and control group (Table 1). Majority of participants in this study were Kuwaiti in nationality contributing 73.7% and 84.2% in vitiligo and control groups, respectively. Whereas, 26.3% were Non-Kuwaiti participants in Vitiligo group and only 15.8% in control group (Table 1).
Vitiligo
Control
Chi-square value
P-value
n
%
n
%
Gender
M
6
31.6
10
52.6
1.727
0.189
F
13
68.4
9
47.4
Total
19
100
19
100
Age
Min-max
20-64
19-75
0.191
Median
38
29
IQR
25-47
20-45
Sum of reads
415.5
325.5
Nationality
K
14
73.7
16
84.2
0.693
NK
5
26.3
3
15.8
Total
19
100
19
100
Table 1: Demographic data of vitiligo and control group.
Vitamin D is further categorized as either; Deficient, ranging 0-49 nmol/L; Insufficient, that is 50-74 nmol/L; Sufficient, 75-125 nmol/L; Ideal, 126-175 nmol/L and Toxic >375 nmol/L. upon this classification, the results have shown slight difference between vitamin D level in case and control group (Figure 1).
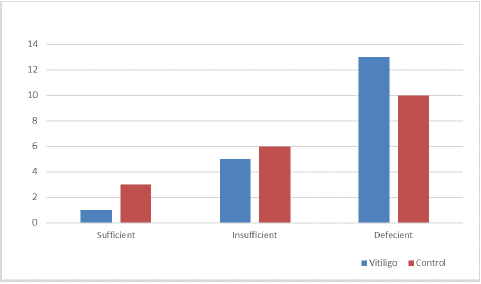
Figure 1: Vitamin D categories between Vitiligo and Control group.
We anticipated the results of vitamin D in both groups using Mann-Whitney test which revealed a lower in control group 37 (21-56) than that in vitiligo group 49 (31-72). The sum of reads were 328.5 and 412.5 in vitiligo and control group, respectively. With an exact P-value of 0.223, indicating non-significant data was evaluated.
Vitamin D
Vitiligo
Control
Min-Max
8-111
20-110
Median
37
49
IQR
21-56
31-72
Sum of reads
328.5
412.5
Exact P-value
0.223
Table 2: Mann-Whitney Test on Vitamin D level in Vitiligo and control group.
Among vitiligo cases, more than two-thirds (68.4%) were receiving topical treatment, while the remaining (31.6%) were treated by both laser and topical treatment (Figure 2).
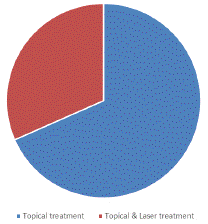
Figure 2: Treatment modality received in patients with Vitiligo.
There are higher percentages among Vitiligo cases who had past medical history of hypertension, while the control group who had past medical history of acne vulgaris and psoriasis. The differences were statistically significant, where p-value was <0.001 (Table 3).
Past Medical History
Vitiligo Group
Control Group
n
%
n
%
Acne Vulgaris
0
0.00
12
63.16
Bronchial Asthma
1
5.26
0
0.00
Celiac disease
1
5.26
0
0.00
Dermatitis
0
0.00
1
5.26
Hypertension
3
15.79
0
0.00
Hyperprolactinemia
1
5.26
0
0.00
Inflammatory Bowel Disease
1
5.26
0
0.00
Melasma
0
0.00
1
5.26
None
10
52.63
0
0.00
Pruritis
0
0.00
1
5.26
Psoriasis
2
10.53
4
21.05
Likelihood Ratio
45.041
p
<0.001
Table 3: Comparison between cases and control regarding past medical history.
Discussion
There is a widespread deficiency in vitamin D, and research conducted in 2008 indicated that this deficiency may constitute a global pandemic [16]. In this study, the median level of total 25-hydroxyvitamin D among vitiligo patients was 37, whereas it was 49 in the control group, with no discernible difference from a normal level (P=0.223). There was no significant difference between age groups participating in the study regarding total vitamin D level. In general, there was no statistically significant difference between the mean levels of total 25-hydroxyvitamin D in the male and female subjects in this study. Of course, inadequate sun exposure and a lack of foods containing vitamin D are contributing factors to low vitamin D serum levels [17]. Since many people in our region have darker skin tones, the dark skin of our group may also be a contributing factor. In addition, pigmented skin is known to increase the risk of vitamin D insufficiency because melanin filters UV rays. Vitamin D deficiency is extremely common in our population [18], and one explanation for this deficiency may be related to the more conservative clothing style (for cultural and religious reasons), particularly in females wearing the hijab and covering their bodies almost entirely, which may prevent the synthesis of vitamin D.
We did our retrospective study to observe the significant role of vitamin D deficiency with the occurrence and the severity of getting vitiligo disease, which will help to control and improve the quality of life for vitiligo patients, in the end of our study we found out that there was almost no difference in age and gender between vitiligo and control group, and the results indicated that non-significant data were obtained.
As far as we know, vitamin D has a big impact on keratinocytes and melanocytes. According to research, vitamin D inhibits the expression of interleukin (IL)-6, IL-8, Tumor Necrosis Factor (TNF) alpha, and TNF-gamma by doing this, it has immunomodulatory effects [19]. It has been reported that repigmentation takes place in vitiligo skin lesions with the help of vitamin D3 by increasing tyrosinase activity and melanogenesis in vitro [20]. The results of this study could be limited by the relatively small number of patients and controls. Some patients were excluded due to missing data while others were due to comorbidities such as diabetes mellitus and other diseases, and others due to age. High prevalence of vitamin D deficiency in this population is another possible limitation to this study.
Conclusion
According to our research, vitamin D levels in the case group were slightly lower than the control group. To acquire a greater understanding of the subject, additional research on other types of vitiligo with varying degrees of severity, using larger sample sizes and longer time periods, is necessary. To thoroughly evaluate vitamin D's effectiveness in treating vitiligo, we advise using it as a treatment. Additional research is necessary to make this recommendation.
References
- NIAMS. Systemic lupus erythematosus (lupus) [internet]. National Institute of Arthritis and Musculoskeletal and Skin Diseases. 2019.
- Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014; 21: 319-29.
- Roffe-Vazquez DN, Huerta-Delgado AS, Castillo EC, Villarreal-Calderón JR, Gonzalez-Gil AM, Enriquez C, et al. Correlation of vitamin D with inflammatory cytokines, atherosclerotic parameters, and lifestyle factors in the setting of heart failure: A 12-month follow-up study. Int J Mol Sci. 2019; 20: 5811.
- Vitamin D. South Tees Hospitals NHS Foundation Trust. South Tees Hospitals NHS Foundation Trust.
- Al Ghamdi K, Kumar A, Moussa N. The role of vitamin D in melanogenesis with an emphasis on vitiligo. Indian J Dermatol Venereol Leprol. 2013; 79: 750-8.
- Mahmmod Z, Ismael DK. Vitamin D deficiency in patients with vitiligo: A cross-sectional study from Basrah, Iraq. Cureus. 2021; 13: e20733.
- Beheshti A, Ghadami H, Barikani A, Haj Manouchehri F. Assessment of vitamin D plasma levels in patients with vitiligo vulgaris. Acta Med Iran. 2014; 52: 601-6.
- Alshiyab DM, Al-Qarqaz FA, Heis LH, Muhaidat JM, Eddin WS, Atwan AA. Assessment of Serum Vitamin D Levels in Patients with Vitiligo in Jordan: a Case-Control Study, [Eddin WS, Atwan AA]. Dermatol Res Pract. 2019; 2019: 2048409.
- Song J, Liu K, Chen W, Liu B, Yang H, Lv L, et al. Circulating vitamin D levels and risk of vitiligo: evidence from meta-analysis and Two-Sample Mendelian randomization. Front Nutr. 2021; 8: 782270.
- Varikasuvu SR, Aloori S, Varshney S, Bhongir AV. Decreased circulatory levels of vitamin D in vitiligo: a meta-analysis. An Bras Dermatol. 2021; 96: 284-94.
- Kechichian E, Ezzedine K. Vitamin D and the skin: an update for dermatologists. Am J Clin Dermatol. 2018; 19: 223-35.
- Holick MF. Vitamin D status: Measurement, Interpretation, and clinical application. Ann Epidemiol. 2009; 19: 73-8.
- Souberbielle JC, Body JJ, Lappe JM, Plebani M, Shoenfeld Y, Wang TJ, et al. VitaminDand musculoskeletal health, cardiocascular disease, autoimmunity and cancer: recommendations for clinical practice. Autoimmun Rev. 2010; 9: 709-15.
- Dawson-Hughes B, Heaney RP, Holick MF, Lips P, Meunier PJ, Vieth R. Estimates of optimal vitamin D status. Osteoporos Int. 2005; 16: 713-6.
- Vieth R. Why the minimum desirable serum 25-hydroxyvitamin D level should be 75 nmol/L (30 ng/mL). Best Pract Res Clin Endocrinol Metab. 2011; 25: 681-91.
- Moradzadeh K, Larijani B, Keshtkar AA, Hossein-Ne A, Rajabian R, Nabipour I, et al. Normative values of vitamin D among Iranian population: A population based study. Int J Osteoporos Metab Disord. 2007; 1: 8-15.
- Lips P. Vitamin D status and nutrition in Europe and Asia. J Steroid Biochem Mol Biol. 2007; 103: 620-5.
- Zhang FF, Al Hooti S, Al Zenki S, Alomirah H, Jamil KM, Rao A, et al. Vitamin D deficiency is associated with high prevalence of diabetes in Kuwaiti adults: results from a national survey. BMC Public Health. 2016; 16: 100.
- Koizumi H, Kaplan A, Shimizu T, Ohkawara A. 1, 25-dihydroxyvitamin D3 and a new analogue, 22-oxacalcitriol, modulate proliferation and interleukin-8 secretion of normal human keratinocytes. J Dermatol Sci. 1997; 15: 207-13.
- Deleuran B, Abraham DJ. Possible implication of the effector CD4+ T-cell subpopulation TH17 in the pathogenesis of systemic scleroderma. Nat Clin Pract Rheumatol. 2007; 3: 682-3.